Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
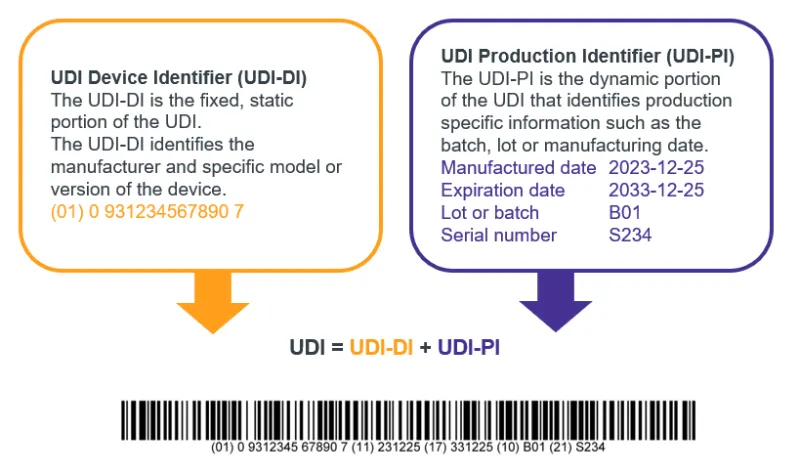
A UDI identifies a specific model and production run of a medical device.
It has 2 parts:
- UDI Device Identifier (UDI-DI)
- UDI Production Identifier (UDI-PI).
The full UDI links a model to its production run and is used to trace the model of device down to the batch, lot or manufacturing date.
If a recall affects one production run, the UDI-DI identifies the model, and the UDI-PI identifies the affected batch.