Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
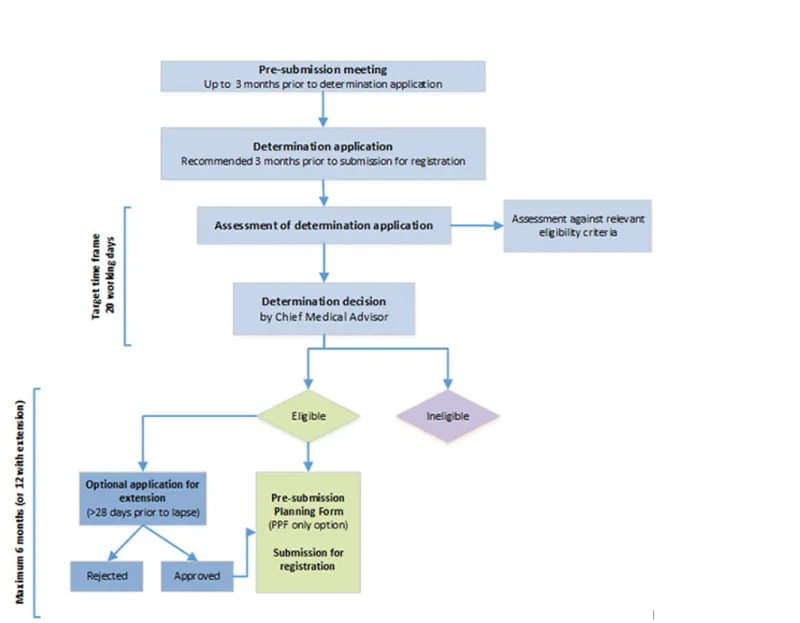
This guidance is for sponsors intending to use the provisional approval pathway for prescription medicines. This pathway allows a time-limited provisional registration on the Australian Register of Therapeutic Goods (ARTG).
This guidance assists sponsors with the process of applying for a provisional determination, which is the first step of the provisional approval pathway.