Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
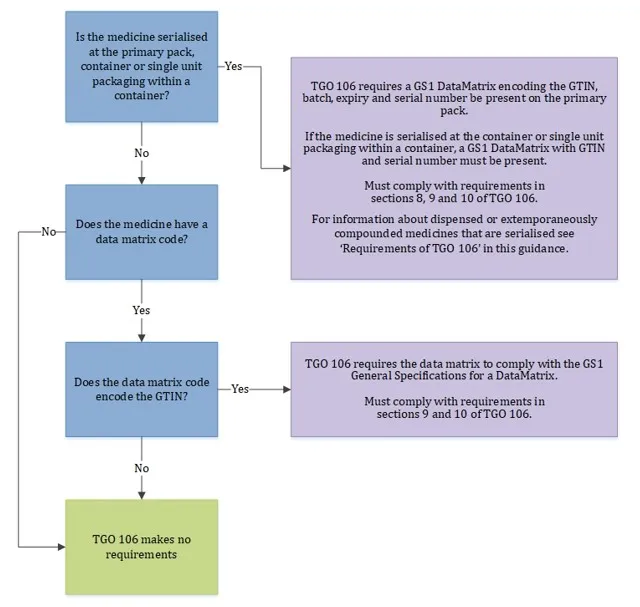
This guidance is for sponsors and manufacturers supplying medicines in Australia that are serialised or have data matrix codes. It describes the requirements in the Therapeutic Goods (Medicines – Standard for Serialisation and Data Matrix Codes) (TGO 106) Order 2021.
To understand the meaning of terms used in this guidance refer to: