One of our functions is to monitor and enforce compliance with laws that regulate therapeutic goods in Australia. This page outlines how we approach and manage non-compliance.
Regulatory compliance framework
We safeguard the health of the Australian community through effective and timely regulation of therapeutic goods. We do this by monitoring and enforcing compliance with the legislation and regulations for the import, export, manufacture, advertising, and supply of therapeutic goods. This work helps ensure that the medicines and medical devices that millions of Australians rely on are safe and fit for purpose.
Our compliance functions support the broader regulatory objectives, including consumer protection, and enabling a fair market for industry.
The work we do is guided by the following key principles.
- Encouraging voluntary compliance through education.
- Prioritising our compliance and enforcement activities based on risks to public health and safety, and confidence in our regulatory framework.
- Ensuring our actions are evidence-based and proportionate to the seriousness of the entity’s behaviour and approach to their compliance obligations.
- Showing our commitment to transparency by reporting on enforcement actions and compliance outcomes.
We can take action in relation to therapeutic goods and/or products. However, we aren’t responsible for things such as:
- health services (except in relation to therapeutic goods)
- medical practitioner conduct
- drug offences that are investigated by police
- general consumer goods.
We may refer:
- any practice concerns to state or territory health departments and/or the Australian Health Practitioner Regulatory Agency (Ahpra).
- pricing issues to the Australian Competition and Consumer Commission (ACCC) or state or territory fair trading agencies.
Education
We educate industry, health professionals and advertisers to promote voluntary compliance with our regulatory requirements.
We provide information that:
- raises awareness of legal obligations
- helps industry understand how to comply and meet their obligations
- keeps industry updated on changes to the regulatory requirements
- empowers consumers and health professionals to recognise non-compliant products and advertising.
We provide education through a range of channels as outlined in the Therapeutic goods import, advertising and supply compliance education strategy.
Prioritisation
Detection and assessment
We use a combination of monitoring strategies to support our compliance programs. Our monitoring programs are both proactive and responsive, including acting upon signals and reports of non-compliance. These reports come from internal and external sources, including members of the public via our report a breach page.
Reports made to us are held in strict confidence and are also able to be sent in anonymously.
Assessment and case selection
Due to the volume of reports and complaints received we cannot pursue all matters that come to our attention. We focus on those matters that concern public safety, allege serious breaches of the Therapeutic Goods Act 1989 (the Act) and regulations, or involve repeated or wilful non-compliance.
All signals of non-compliance are recorded and considered. Once received, a report is assessed by considering:
- if it relates to something we regulate and can take action on
- the risk of harm to consumers related to a product, including the way in which a product is used or promoted for use
- the nature of the breach
- the vulnerability of the target market
- the size, reach and compliance history of the entity or industry sector.
We use this risk assessment to determine the priority of the matter and how it will be actioned.
For example, a report may be immediately investigated, recorded for intelligence purposes, or referred for investigation later. This risk assessment and these factors also are considered for determining the action we will take.
Compliance and enforcement actions
Our response to non-compliance takes into account the risk and the nature of the breach. It is determined on a case-by-case basis.
A range of compliance and enforcement actions are used to address alleged non-compliance. These actions are shown in the diagram below and are used either individually or in combination.
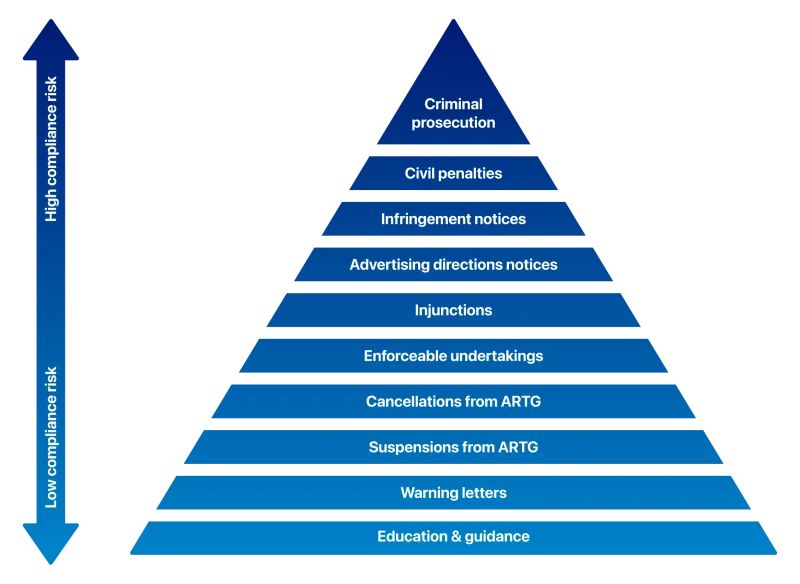
You can find more information on the compliance actions available to us at Compliance actions and outcomes.
Transparency
We measure and report on our performance to ensure accountability and continued improvement. As part of our commitment to transparency about our compliance actions, we publish:
- outcomes from our Compliance actions and outcomes
- performance information, via our annual reports
- media releases
- key actions related to Unlawful activities surrounding therapeutic goods.
We do not comment on matters under investigation. However, we may make a public statement on an ongoing matter where it is in the public interest or necessary for the investigation.
Managing regulatory compliance
Further information on specific monitoring and compliance functions can be accessed from the links below.
Compliance principles and priority focus areas
Vaping goods
Information on compliance and enforcement activities related to vaping goods is available on the dedicated vaping hub.
Medicines
- Medicine shortages in Australia: reporting obligations and the TGA's compliance framework
Guidance on the mandatory reporting scheme for medicine shortages and permanent discontinuations of supply of mostly prescription medicines. - Listed medicine compliance reviews
If we find that a medicine is non-compliant, we take action according to the risk posed by the medicine to ensure the medicine is either brought into compliance or removed from the ARTG. - Pharmacovigilance Inspection Program
Our Pharmacovigilance Inspection Program (PVIP) has been implemented as an initiative to help sponsors of medicines to meet their pharmacovigilance obligations.
Medical devices
If we find a safety or performance issue with a medical device, we take action based on the risk it poses. We ensure the device is either brought into compliance with the Essential Principles or removed from the ARTG. More information on the Essential Principles for medical devices.
We monitor the regulatory compliance of software and AI medical devices. This may include targeted action in response to alleged non-compliance.
Manufacturing therapeutic goods
- Conformity assessment certification
Conformity assessment is the systematic and ongoing examination of evidence and procedures to ensure that a medical device (including IVD medical devices) complies with the Essential Principles, which set out fundamental safety and performance requirements. - Good manufacturing practice - an overview
Good Manufacturing Practice (GMP) describes a set of principles and procedures that when followed helps ensure that therapeutic goods are of high quality. - Guidance on the management of GMP compliance signals
Outlines the GMP compliance requirements (according to the Manufacturing Principles) for manufacturing biologicals and medicines intended for supply in Australia. It also describes our framework for managing GMP compliance signals.
Laboratory testing
- Testing of therapeutic goods
Our laboratories test therapeutic goods for quality. A risk-based process is applied to determine how products are tested.
Page history
Added link to Key actions related to unlawful activities surrounding therapeutic goods.
- Removed link to Compliance Priorities 2023-25
- Added Compliance Principles 2026 and 2027
Original publication.
Added link to Key actions related to unlawful activities surrounding therapeutic goods.
- Removed link to Compliance Priorities 2023-25
- Added Compliance Principles 2026 and 2027
Original publication.


