Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
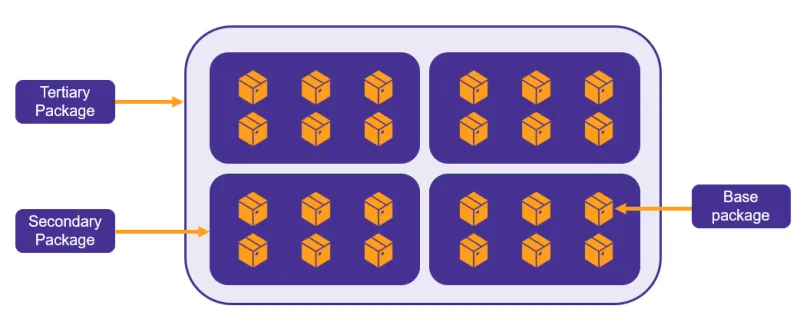
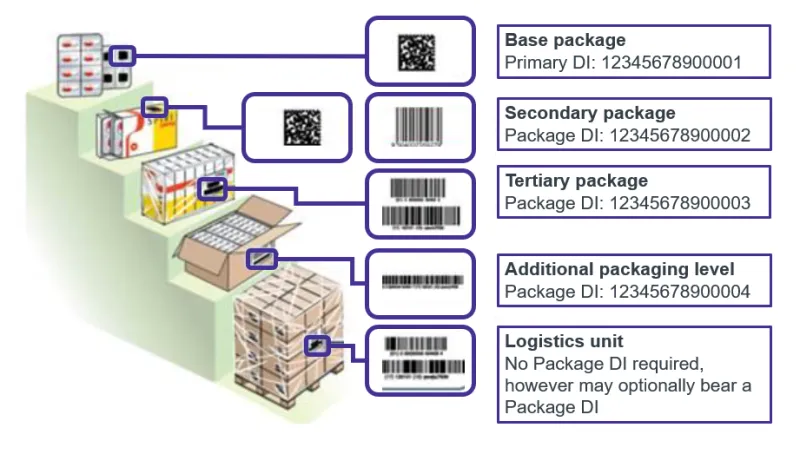
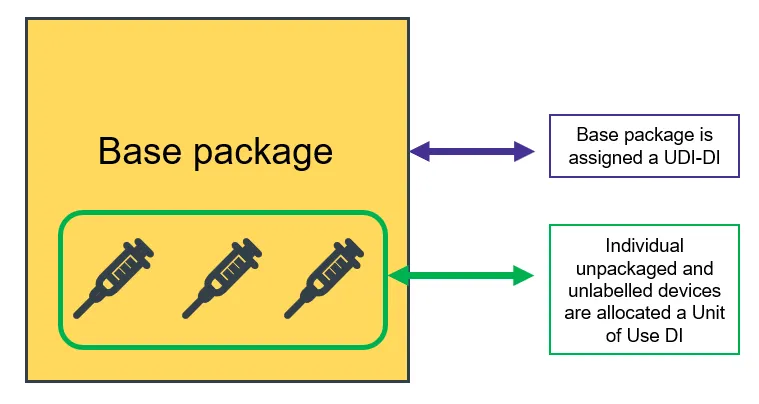
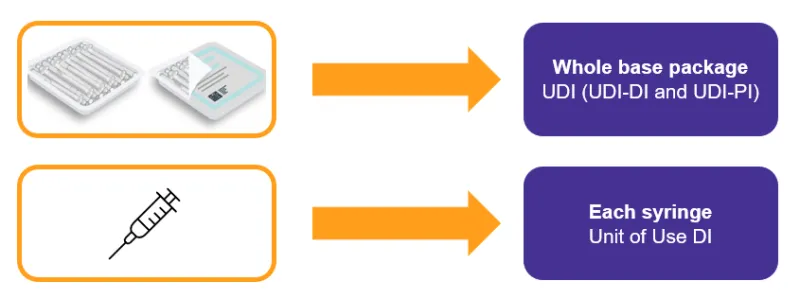
As a manufacturer, you must apply a UDI to:
- the device label
- the device itself, if direct marking applies
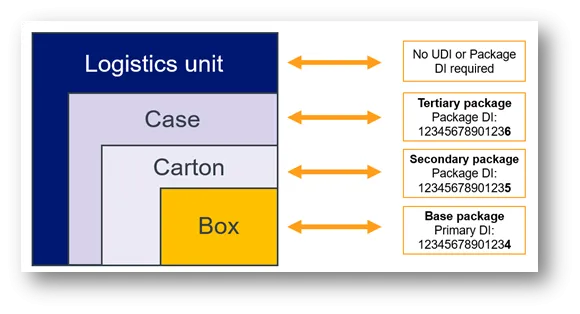
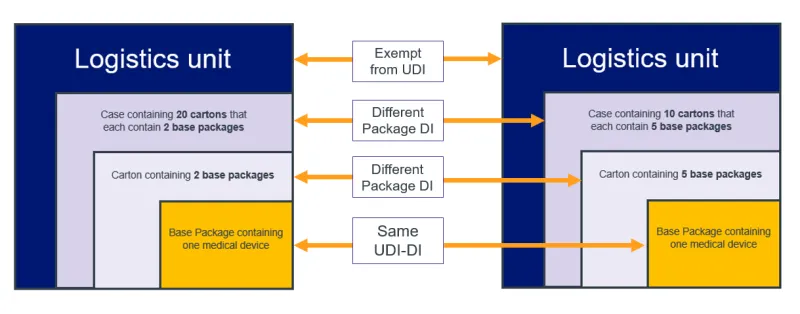
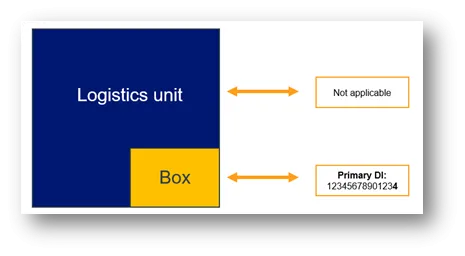
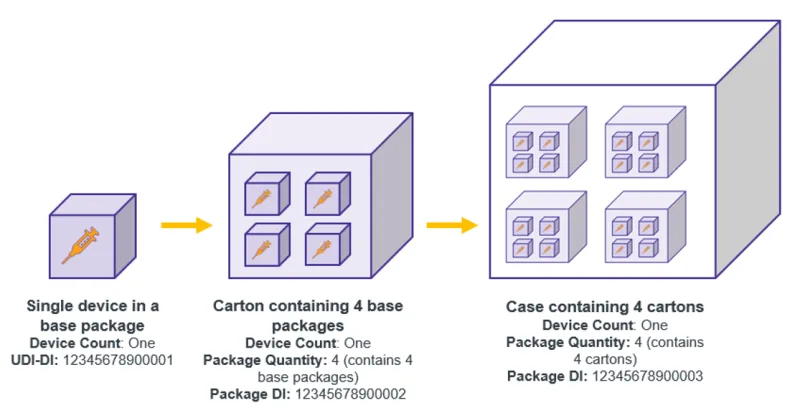
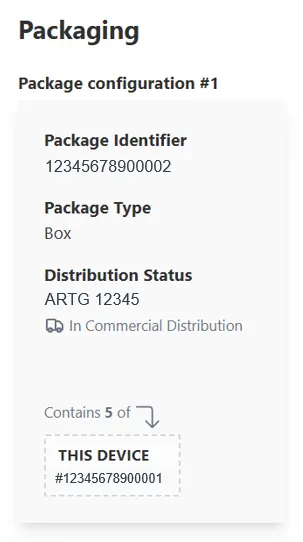
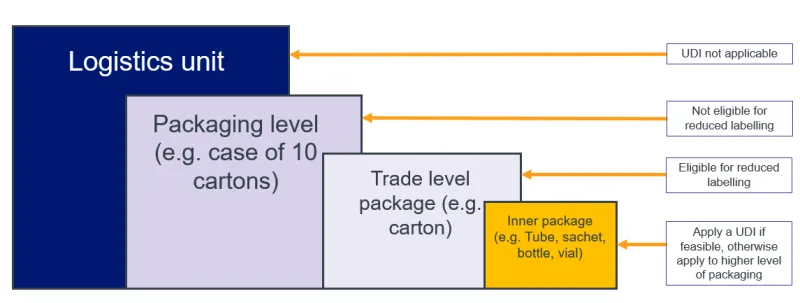
- all applicable higher levels of packaging
- Patient Implant Cards (PICs), if applicable.
UDIs are applied in the form of a UDI Carrier.
UDI Carrier
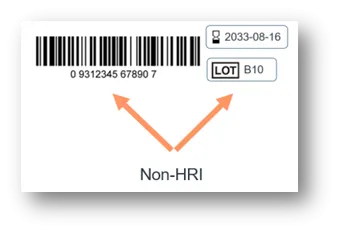
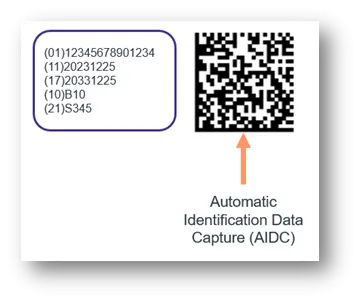
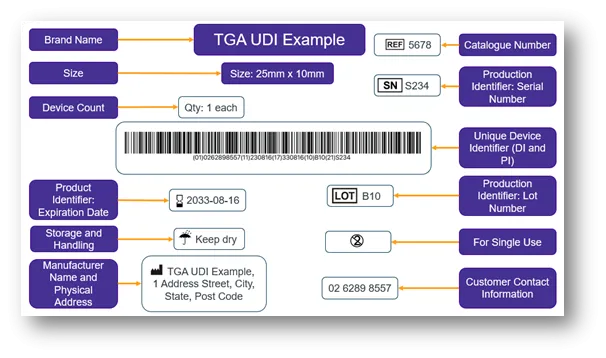
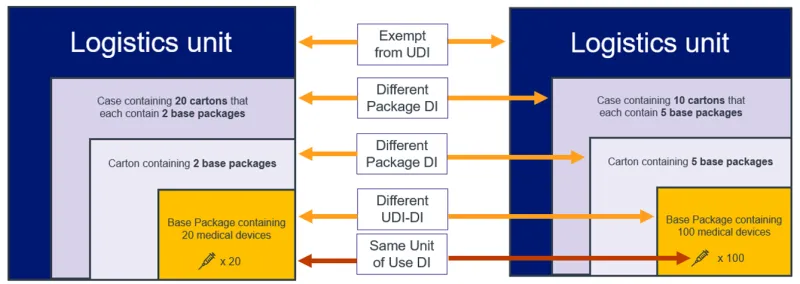
The UDI Carrier is the physical means to convey the UDI. The UDI Carrier displays the full UDI (UDI-DI and UDI-PI) in 2 forms:
- Human Readable Interpretation (HRI)
- Automatic Identification Data Capture (AIDC).
This is to ensure the device can be both read by humans and machines.
The image below demonstrates how the UDI-DI plus the UDI-PI form the full UDI, and how the full UDI in HRI and AIDC form the UDI Carrier.