Recently published
This page was published on [date_placeholder].
Recently updated
This page was updated on [date_placeholder]. See page history for details.
Purpose
Platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and conditioned serum are products made from human blood that have been concentrated with the aim to enrich for growth factors and anti-inflammatory compounds (cytokines).
These products are normally used in an "autologous" manner, which means the blood is first collected from a patient, processed in the laboratory, and then returned to the same patient. Commercially-supplied kits and equipment are often used for the processing of the collected human blood.
This guidance should be used to determine whether a particular PRP, PRF or conditioned serum product is being manufactured and supplied for a therapeutic use, and, where it is, the level of TGA regulation that is applicable.
The introduction of TGA's Biologicals Regulatory framework in 2011 and subsequent amendments to the regulation of certain human cell and tissue products have not had a significant impact on the regulation of these products.
Legislation
Is my product PRP, PRF or conditioned serum?
The major differences between PRP, PRF and conditioned serum in their preparation from human blood are outlined below. Please note that the diagrams are representative only and individual protocols vary.
Platelet-rich plasma (PRP)
PRP is prepared from blood collected into a syringe containing an additive (anticoagulant) to prevent the blood from clotting.
In order to isolate the liquid, cell-free plasma, the blood tube is generally centrifuged to separate the red blood cells (RBC; erythrocytes), with the platelets and white blood cells (WBC) remaining in the upper layers of the plasma.
The next steps to collect the PRP layer vary between individual protocols but are intended to discard the plasma layer not containing platelets (PPP; platelet-poor plasma) and leave the PRP layer.
Preparation of platelet-rich plasma (PRP)
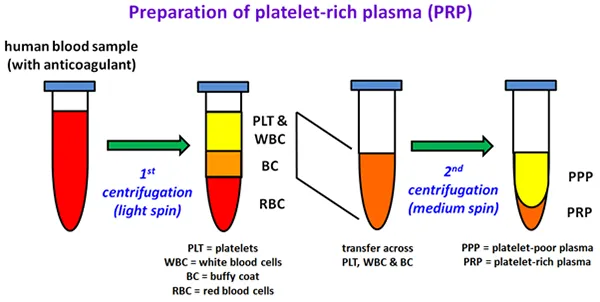
Diagram illustrating the preparation of platelet-rich plasma (PRP) in four steps, with four test tubes shown side by side:
- initial test tube labelled "human blood sample (with anticoagulant)" containing red liquid
- after first centrifugation (light spin), the second test tube shows layered contents: top yellow layer labelled "PLT & WBC" (platelets and white blood cells), thin middle layer labelled "BC" (buffy coat), bottom red layer labelled "RBC" (red blood cells)
- transfer of upper layers to new test tube, leaving behind red blood cell layer
- after second centrifugation (medium spin), the final test tube shows two layers: upper larger layer labelled "PPP" (platelet-poor plasma), bottom smaller yellow layer labelled "PRP" (platelet-rich plasma).
Green arrows indicate progression between each of the four steps, with abbreviations listed below the diagram.
Platelet-rich fibrin (PRF)
PRF is a second-generation PRP where platelets and WBC form a fibrin matrix (scaffold) that claims to localise growth factors and increase their concentration.
Like PRP, PRF is prepared from a blood collection, but no anticoagulant is added, and the blood is swiftly centrifuged after collection (≤ 2 minutes) in order to isolate the fibrin matrix.
After centrifugation, the upper layer contains PPP, the PRF matrix is in the middle layer, with RBC at the bottom.
Preparation of platelet-rich fibrin (PRF)
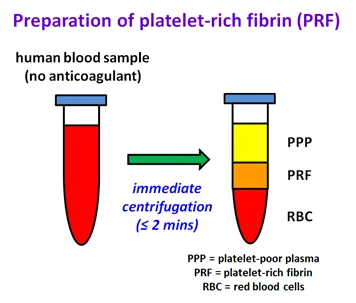
A diagram illustrating the preparation of platelet-rich fibrin (PRF), with two test tubes shown side by side.
The first test tube on the left depicts a human blood sample without anticoagulant, red in colour.
An arrow pointing from the first test tube to the second test tube is labelled 'immediate centrifugation (≤ 2 mins)'.
The second test tube on the right-hand side shows the sample with three distinct layers after centrifugation:
- a yellow layer at the top labelled platelet-poor plasma (PPP)
- a middle orange layer labelled platelet-rich fibrin (PRF)
- a red layer at the bottom labelled red blood cells (RBC).
Conditioned serum
Like PRP and PRF, conditioned serum is prepared from a blood collection, but the blood is allowed to naturally clot (no anticoagulant).
In order to isolate the liquid serum, the blood tube is centrifuged to separate the blood clot. The serum fraction does not contain platelets.
In contrast to PRP and PRF, which uses centrifugation to separate the blood components, the blood tube is then incubated for a period of time in the laboratory to allow for full activation of WBC and the secretion of cytokines into what is known as conditioned serum.
Preparation of conditioned serum
Preparation of conditioned serum
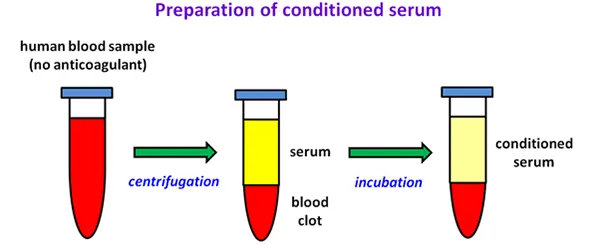
Diagram showing three stages of preparing conditioned serum from human blood.
Stage 1: A test tube contains a red human blood sample without anticoagulant.
Stage 2: After centrifugation, the tube shows yellow serum above a red blood clot.
Stage 3: After incubation, the final tube contains pale yellow conditioned serum above the red blood clot.
Green arrows indicate the progression between stages, labelled 'centrifugation' and 'incubation' respectively.
Is my PRP, PRF or conditioned serum product a therapeutic good?
One of the main factors in determining whether a product is a cosmetic or a biological is the claims made about the product.
Even if a product is intended for marketing as a cosmetic or orthopaedic purpose, it may still be classified as a therapeutic good and subject to regulation by TGA. This depends on:
- its ingredients
- its route of administration
- if therapeutic claims are made on its label or in advertising.
For example, PRP, PRF or conditioned serum injected for treating a condition such as osteoarthritis is likely to be regulated as a therapeutic good since this is a therapeutic use.
Where products have a therapeutic use, they would normally be regulated by TGA, subject to any applicable exclusions or exemptions.
If my PRP, PRF or conditioned serum product is a therapeutic good, how will it be regulated?
Where PRP, PRF or conditioned serum are collected, manufactured and administered by a medical practitioner to a patient under their care, exemptions are likely to apply. See below for more detail on the provisions that must be met for the exemptions to apply, and other pathways these goods may be supplied.
Excluded from regulation
When the blood is collected from a patient who is under the clinical care of a medical or dental practitioner and the subsequent PRP, PRF or conditioned serum product is manufactured and used within an accredited hospital, this product is likely to be excluded from regulation.
Please note that this pathway only applies where the products are not advertised to consumers.
Exempt from regulation
Most use of PRP, PRF or conditioned serum products is likely to be regulated as blood components, but exempt from most regulation requirements when all of the following criteria are met:
- collected from a patient who is under the clinical care of a medical or dental practitioner registered under a law of a state or an internal territory
- manufactured and used by the practitioner with primary responsibility for clinical care OR by a person or persons under the professional supervision of that practitioner
- for a single indication in a single clinical procedure
- are minimally manipulated and for homologous use.
It should be noted that:
- There are substantial variations in the preparation methods and level of processing used to manufacture these products, so the level of regulation applicable should be carefully considered.
- These exemptions only apply when the PRP, PRF or conditioned serum product is manufactured and administered by, or under the supervision of, a registered medical or dental practitioner for a patient under their care. This exemption does not apply to other health practitioners.
- The intended clinical use of the PRP, PRF or conditioned serum product under these exemptions should still be justified based on proven evidence of safety and efficacy.
- Where one or more criteria are not met, the exemptions from certain regulatory requirements do not apply.
- Where equipment such as a commercially-supplied kit is used in the manufacture of a PRP, PRF or conditioned serum product, it may also be subject to regulation as a medical device.
If the PRP, PRF and conditioned serum product is subject to more than minimal manipulation, it may be subject to a higher level of regulation as a biological rather than a blood component.
For example, where the PRP, PRF or conditioned serum product is prepared using any compounds that alter the plasma, serum, or other blood components (e.g. coatings on a blood tube that activate a blood component), this would be considered more than minimal manipulation of the blood and subjected to higher regulation.
Similarly, if the PRP, PRF or conditioned serum product is cultured in the incubator with other cells, tissues, growth factors, or other ingredients prior to use, the products are unlikely to meet the definition of PRP, PRF or conditioned serum and could then be subjected to regulation as a biological.
For further information on the regulation of biologicals, please refer to the Australian regulatory guidelines for biologicals (ARGB).
Can I advertise my PRP, PRF or conditioned serum product?
Advertising of PRP, PRF or conditioned serum products to the public is not prohibited (unless supply is through the excluded goods provision), but advertising must comply with the requirements of the Act and the Therapeutic Goods Advertising Code (the Code).
Are commercially-supplied kits and equipment used to prepare PRP, PRF or conditioned serum subject to regulation?
Independent of whether a PRP, PRF or conditioned serum product is subject to regulation as blood component (medicine) or biological, the kits or equipment used to prepare the product may be regulated as medical devices.
Page history
Title changed from 'Regulation of platelet-rich plasma (PRP), platelet-rich fibrin (PRF) and conditioned serum' to 'Regulating platelet-rich plasma (PRP), platelet-rich fibrin (PRF) and conditioned serum' as part of migration to new 'Guidance' content type:
- Consistent ‘Purpose’ heading.
- ‘Legislation’ section to clearly show which laws the Guidance relates to.
- ‘Page history’ section replaces document version history.
- New page navigation features.
- Updated page summaries.
- Complex images include long descriptions.
- New ‘Save as PDF’ feature.
Title changed from 'Regulation of platelet-rich plasma (PRP), platelet-rich fibrin (PRF) and conditioned serum' to 'Regulating platelet-rich plasma (PRP), platelet-rich fibrin (PRF) and conditioned serum' as part of migration to new 'Guidance' content type:
- Consistent ‘Purpose’ heading.
- ‘Legislation’ section to clearly show which laws the Guidance relates to.
- ‘Page history’ section replaces document version history.
- New page navigation features.
- Updated page summaries.
- Complex images include long descriptions.
- New ‘Save as PDF’ feature.

