Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
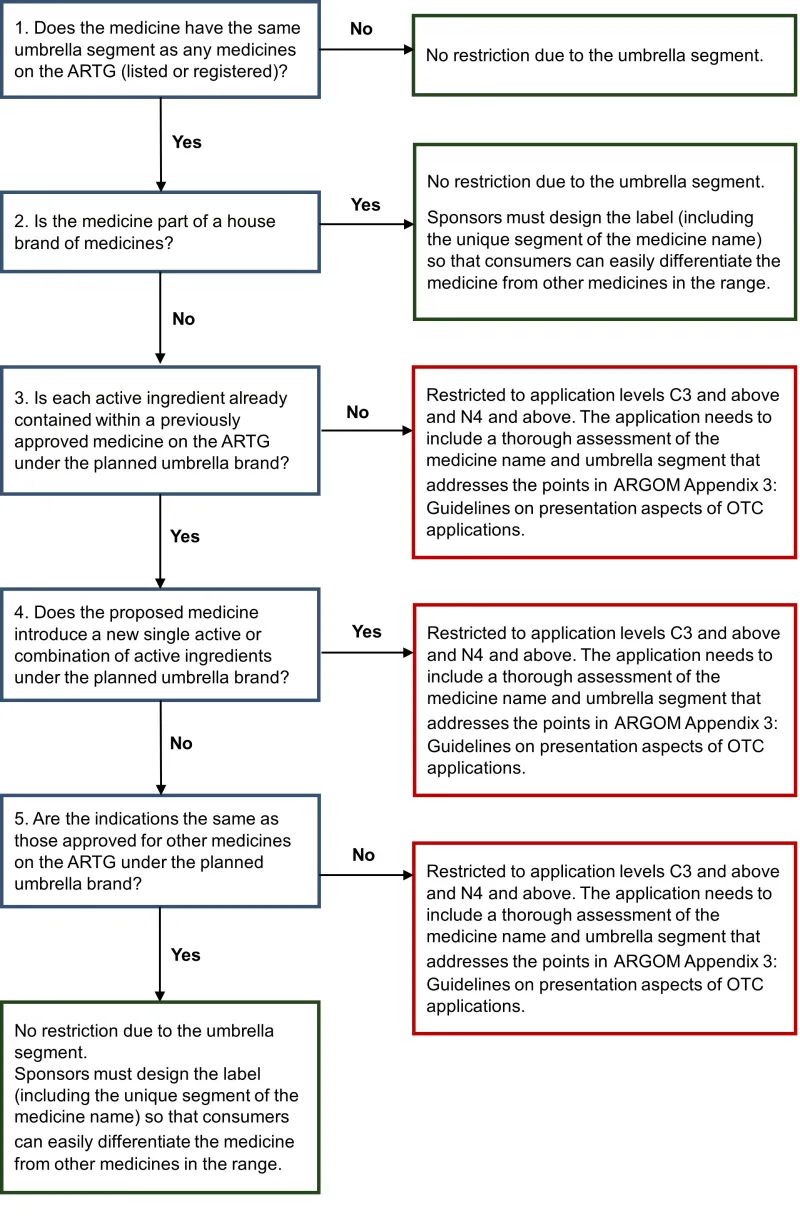
This guidance helps sponsors determine whether their over-the-counter (OTC) medicine application requires a higher level of assessment because of umbrella branding. This is one of the steps required when determining the correct application level for an OTC medicine submission.
When proposing a new name with an umbrella segment, the sponsor needs to:
- consider whether the umbrella segment of the name of their medicine requires a higher level of assessment
- if a higher level of assessment is required, address in the application the relevant considerations outlined in ARGOM Appendix 3: Guidelines on presentation aspects of OTC applications.