Recently published
This page was published on [date_placeholder].
Recently updated
This page was updated on [date_placeholder]. See page history for details.
Purpose
This guidance helps sponsors determine whether their over-the-counter (OTC) medicine application requires a higher level of assessment because of umbrella branding. This is one of the steps required when determining the correct application level for an OTC medicine submission.
When proposing a new name with an umbrella segment, the sponsor needs to:
- consider whether the umbrella segment of the name of their medicine requires a higher level of assessment
- if a higher level of assessment is required, address in the application the relevant considerations outlined in ARGOM Appendix 3: Guidelines on presentation aspects of OTC applications.
What umbrella branding is
Umbrella branding refers to the naming of two or more medicines under the same ‘brand’ name.
The umbrella segment is the part of a medicine name that is used in the name of more than one medicine to create a ‘brand’ for a range of medicines.
Umbrella brands are generally associated with:
- particular actives
- a therapeutic area or set of indications
- a particular sponsor or retailer.
House brand is the term used to describe a range of medicines where the umbrella segment is typically associated only with the sponsor or retailer. These brands or ranges of medicines are not associated with any particular active ingredient(s), or therapeutic area and indications(s), and usually the brand spans a wide range of unrelated medicines.
Possible risks associated with umbrella branding
The presentation of therapeutic goods is unacceptable [Section 3(5), Therapeutic Goods Act 1989] if it is capable of being misleading or confusing as to the content or proper use or identification of the goods and, without limiting the previous words in this subsection, the presentation of therapeutic goods is unacceptable:
- if it states or suggests that the goods have ingredients, components or characteristics that they do not have
For the majority of umbrella branded ranges, such as house brands, the branding does not pose safety or efficacy concerns.
Sometimes, a proposed name with an umbrella segment is capable of being misleading or confusing as to one or more of the following aspects of the medicine:
- content
- proper use
- identification.
In these situations, there is potential for consumers to be confused about the differences between the proposed medicine and the current medicines within that umbrella brand.
Negligible to low risk
If the umbrella segment of a new medicine name poses a negligible to low risk of consumers being misled or confused, then the umbrella segment does not contribute to the determination of the application level.
Examples are when the:
- umbrella segment is a house brand
- medicine is a different strength or dosage form to that previously approved within the umbrella brand.
The risk involved with these applications can be managed by the sponsor designing the label (including the unique segment of the medicine name) so that consumers can easily differentiate the medicine from other medicines in the range.
Higher risk
The suitability of the umbrella segment requires a higher level of assessment for applications that propose an extension of the umbrella brand to:
- new active ingredient(s)
- new therapeutic area(s)
- new indication(s).
The risks related to the extension of the umbrella brand in these situations need to be assessed to ensure that there is no increased safety or efficacy risk to consumers and that the presentation is acceptable. The sponsor needs to address in the application the relevant considerations outlined in ARGOM Appendix 3: Guidelines on presentation aspects of OTC applications.
Higher application levels for higher levels of assessment
The OTC application categorisation framework defines the different OTC medicine application levels and identifies key application criteria. Before making an application, a sponsor needs to determine whether there is a risk associated with an umbrella branding segment that requires a higher level of assessment.
If a higher level of assessment is required, then the application level is restricted to:
- C3 and above for change applications
- N4 and above for new medicine applications.
Once you know if a higher level of assessment is required for the umbrella branding segment, you can determine the appropriate application level for the submission by using:
- the OTC application placement flowchart if you are familiar with the risk-based application category framework
- the OTC application placement question and answer tool if you are not familiar with the risk-based application framework.
Flowchart for determining umbrella branding risk level
Use this flowchart to determine whether the application requires a higher level of assessment because of a risk associated with umbrella branding.
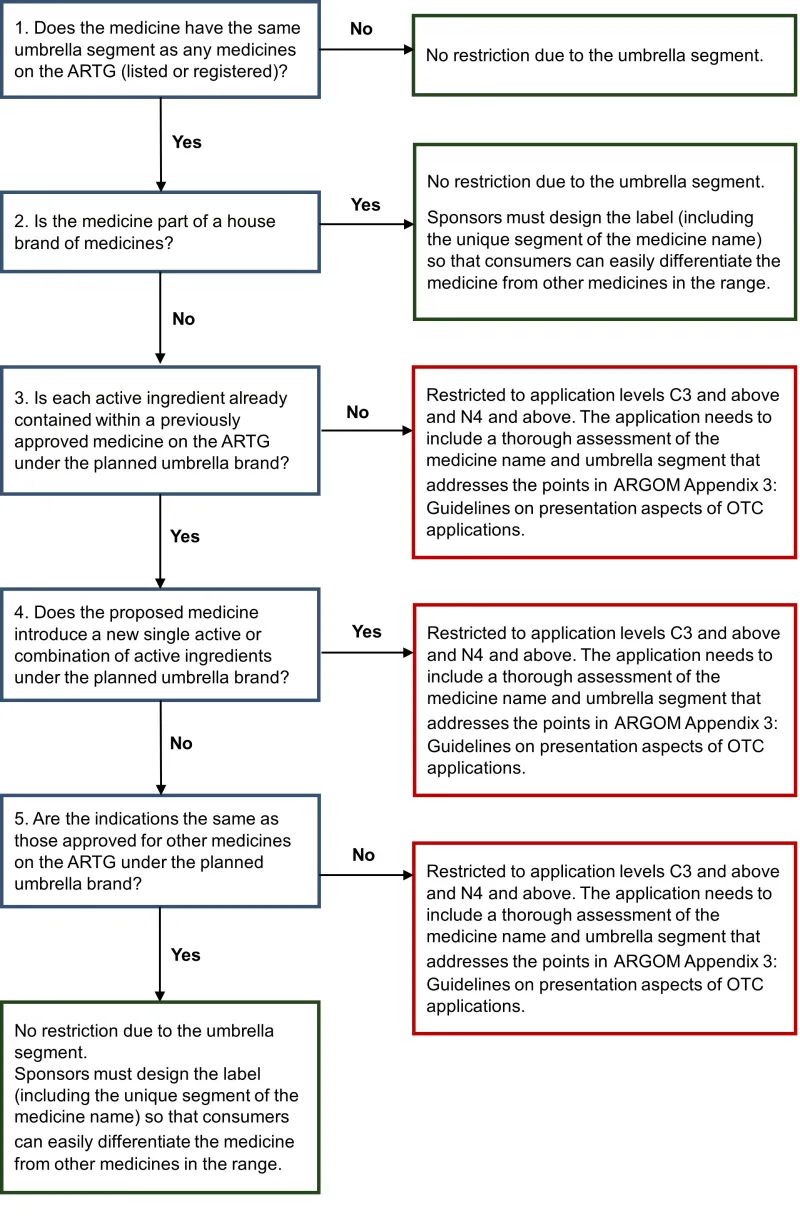
This text representation of the flowchart is a list with numbered steps.
- Does the medicine have the same umbrella segment as any medicines on the ARTG (listed or registered)?
- No: No restriction due to the umbrella segment. End flowchart.
- Yes: Go to step 2.
- Is the medicine part of a house brand?
- Yes: No restriction due to the umbrella segment. Sponsors must design the label (including the unique segment of the medicine name) to ensure consumers can easily differentiate the medicine from other medicines in the range. End flowchart.
- No: Go to step 3.
- Is each active ingredient already contained within a previous approved medicine on the ARTG under the planned umbrella brand?
- No: Restricted to application levels C3 and above and N4 and above. The application needs to include a thorough assessment of the medicine name and umbrella segment that addresses the points in ARGOM Appendix 3: Guidelines on presentation aspects of OTC applications. End flowchart.
- Yes: Go to step 4.
- Does the proposed medicine introduce a new active or combination of active ingredients under the planned umbrella brand?
- Yes: Restricted to application levels C3 and above and N4 and above. The application needs to include a thorough assessment of the medicine name and umbrella segment that addresses the points in ARGOM Appendix 3: Guidelines on presentation aspects of OTC applications. End flowchart.
- No: Go to step 5.
- Are the indications the same as those approved for other medicines on the ARTG under the planned umbrella brand?
- No: Restricted to application levels C3 and above and N4 and above. The application needs to include a thorough assessment of the medicine name and umbrella segment that addresses the points in ARGOM Appendix 3: Guidelines on presentation aspects of OTC applications. End flowchart.
- Yes: Go to step 6.
- No restriction due to the umbrella segment. Sponsors must design the label (including the unique segment of the medicine name) so that consumers can easily differentiate the medicine from other medicines in the range.
- End flowchart.
Page history
- Reworded the guidance
- removed mention of Medsafe and other references to New Zealand.
Content migrated to new guidance content type:
- consistent purpose heading
- legislation section to clearly show which laws the guidance relates to
- page history section replaces document version history
- new page navigation features
- updated page summaries
- complex images include long descriptions
- new ‘Save as PDF’ feature.
- Original publication
- Reworded the guidance
- removed mention of Medsafe and other references to New Zealand.
Content migrated to new guidance content type:
- consistent purpose heading
- legislation section to clearly show which laws the guidance relates to
- page history section replaces document version history
- new page navigation features
- updated page summaries
- complex images include long descriptions
- new ‘Save as PDF’ feature.
- Original publication

