Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
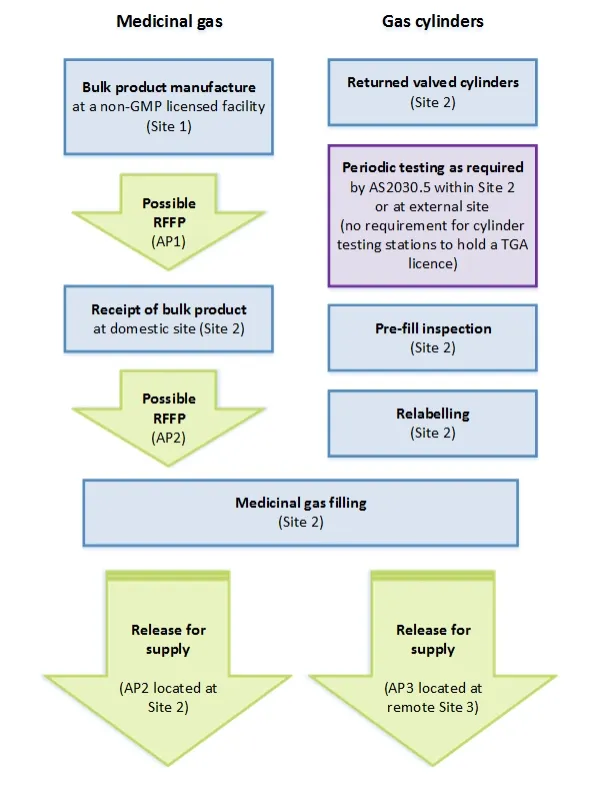
This guidance is for manufacturers of registered medicinal gases. It explains our interpretation and expectations for compliance with specific sections of the PIC/S Guide to Good Manufacturing Practice for Medicinal Products PE009-13 (PIC/S Guide to GMP).
TGA has adopted the PIC/S Guide to GMP, PE009-13 (excluding Annexes 4, 5 and 14), taking effect on 1 January 2018 with a 12-month transition plan.