Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
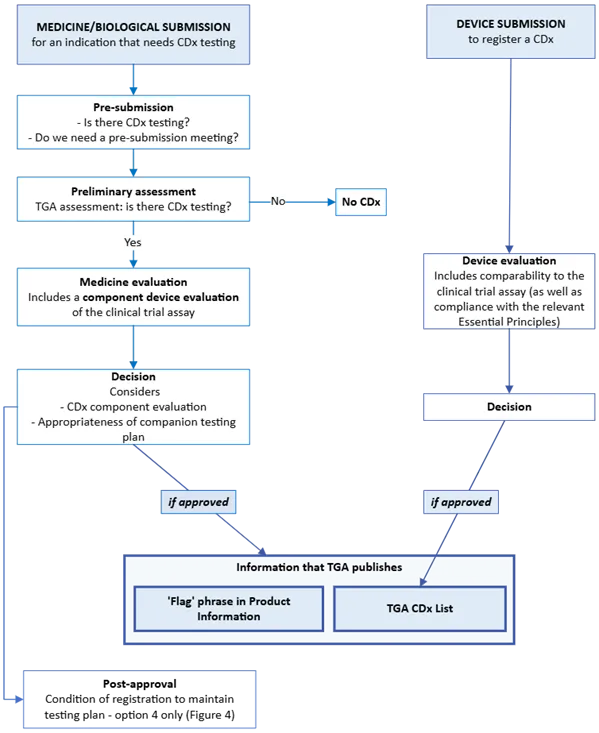
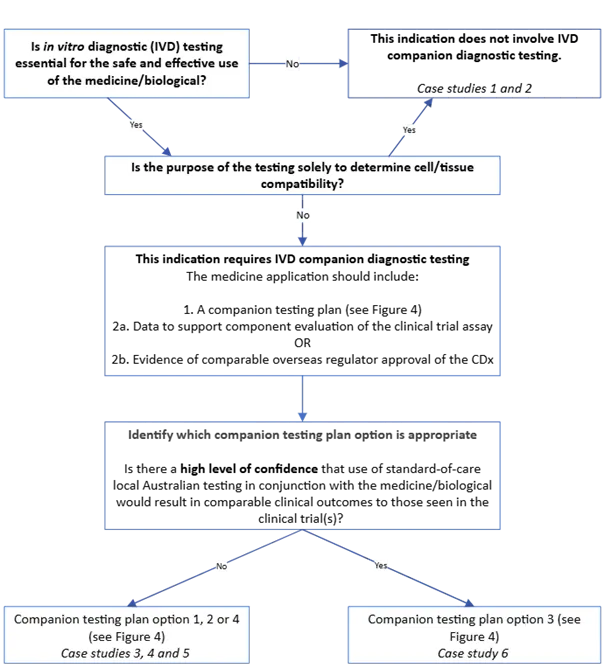
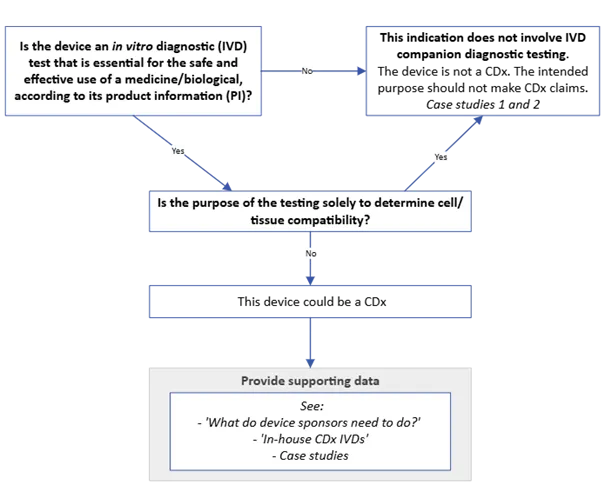
The purpose of this guidance is to outline the regulatory framework for in vitro diagnostic (IVD) companion diagnostics (CDx) and for the medicine or biological that requires CDx testing.
It provides information on clinical and analytical performance requirements for IVD CDx.
The guidance is applicable to both medical device and medicine sponsors.
This guidance addresses requirements for sponsors of commercial IVD CDx and for pathology laboratories with in-house IVD CDx.
However, this guidance does not detail laboratory accreditation requirements for in-house IVD CDx under the National Pathology Accreditation Scheme.
The CDx framework does not restrict the clinical practice of health practitioners prescribing or recommending treatments or diagnostic tests, so this is also not covered in this guidance.
This guidance document does not address the requirements for clinical trials involving IVDs, medicines, or biologicals.
Refer to the clinical trial handbook for guidance on clinical trials.
Reimbursement processes are also not within the scope of this guidance document.
CDx continue to evolve. We will monitor developments, review the implementation of the framework in 12 months’ time and update this guidance as necessary.