Recently published
This page was published on [date_placeholder].
Recently updated
This page was updated on [date_placeholder]. See page history for details.
Purpose
To provide guidance to sponsors, manufacturers and raw material suppliers of the need to undertake a safety assessment to ensure that:
- unprocessed herbal materials for use in complementary medicines; or
- unprocessed herbal materials for use in the production of herbal preparations for use in medicines
that have had their chemical composition modified to the extent that it is significantly different from the original, approved material, are safe for use in complementary medicines.
Scope
This guidance document applies to unprocessed herbal materials whose composition has been modified to the extent that it is significantly different from the original material approved for use in Listed or Registered medicines. Unprocessed (raw) materials of plant origin include the whole, cut, or part of the material, either fresh or dry.
The principles explained in this document may also be applicable to other materials, such as algae or fungi.
For herbal materials that have been further processed, please refer to the Therapeutic Goods Administration (TGA) document 'Guidance on Equivalence of Herbal Extracts in Complementary Medicines'.
Background
The chemical composition of herbal material currently approved for use in Listed or Registered medicines can be altered in a number of ways, including the use of different agricultural practices, selective breeding, the use of recombinant DNA and other techniques.
The resulting modified material may be chemically altered to the extent that its composition is significantly different from the original, approved material.
- The implication of a significant change in chemical composition is that safety (and efficacy) of the modified material can no longer be assured, particularly where it was based upon the safety (and efficacy) of the 'unmodified' material.
- Such changes also have implications for the safety of preparations, such as extracts, that are derived from these materials.
Generally, ingredients that do not comply with a relevant Compositional Guideline, pharmacopoeial monograph, or authoritative source (such as the World Health Organization; or the European Scientific Cooperative on Phytotherapy [ESCOP] monographs), are regarded as separate and distinct and require separate evaluation to determine their eligibility for use in medicines.
A modified herbal material may comply with all the requirements of the Compositional Guideline, pharmacopoeial monograph or authoritative source for the corresponding unmodified herbal material, even though the modification could potentially result in a material with a different safety and / or efficacy profile.
This is because, for some ingredients, especially chemically complex substances, such as herbal materials, it may not be possible for Compositional Guidelines, pharmacopoeial monographs, or authoritative sources, to specify all relevant attributes parameters or the concentration levels for all components, particularly when the majority of components are not chemically identified.
Guidance
Herbal materials approved for use in Listed or Registered medicines, or for use in the production of herbal preparations for use in medicines, are considered separate and distinct if they do not comply with the requirements of the relevant Compositional Guideline, pharmacopoeial monograph, or authoritative source. Such ingredients, or products containing such ingredients, must be approved by the TGA prior to their use in medicines.
For a modified herbal material that does comply with the requirements of the relevant Compositional Guideline, pharmacopoeial monograph, or authoritative source, but which:
- has been modified to the extent that its chemical composition differs from the composition of the herbal material approved for use in Listed or Registered medicines, such that it:
- has one or more components with known therapeutic activity present at greater than +/-10 per cent compared to the amount generally expected1 in the approved material; and / or
- has one or more unidentified components or components not linked with known therapeutic activity, including 'marker compounds', present at greater than +/-20 per cent compared to the amount generally expected in the approved material; or
- contains a chemical component of toxicological significance not generally present, or present in an increased concentration in the approved material; and / or
- results in the removal or reduction in the concentration of a chemical component(s) in the material that contributes to the safety of the approved material2, then the modified material must be assessed for safety prior to use in medicines.
If a modified herbal material meets the above criteria, it is the sponsor's responsibility to ensure that the material is safe prior to its use in medicines. The safety assessment is the responsibility of the sponsor and it may be audited as part of the Office of Complementary Medicines listing compliance review.
If safety cannot be assured, the material must be evaluated by the TGA to determine its eligibility for use in medicines. The accompanying flow chart gives a schematic outline.
Note: Comparison between modified and unmodified ingredients can be made using profile (or fingerprint) chromatograms.
Full details of the principles, development and interpretation of profile chromatograms are included in Australian Regulatory Guidelines for Complementary Medicines (ARGCM) Part III.
Profile chromatograms are particularly useful where the chemical identity of components in an ingredient is unknown or where the components are complex or multitudinous.
Guidance on assessing the safety of an ingredient for use in medicines is given in Part III of the ARGCM.
Flow chart for determining the eligibility of modified unprocessed herbal material for use in complementary medicines
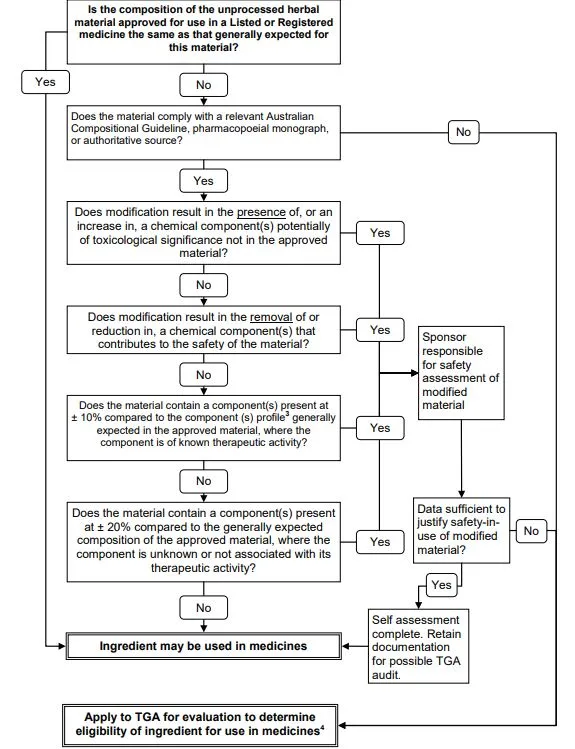
Start:
Is the composition of the unprocessed herbal material approved for use in a Listed or Registered medicine the same as that generally expected for this material?
If Yes: Proceed to next question
If No: Does the material comply with a relevant Australian Compositional Guideline, pharmacopoeial monograph, or authoritative source?
If No: Apply to TGA for evaluation to determine eligibility of ingredient for use in medicines
If Yes: Proceed to next question
Does modification result in the presence of, or an increase in, a chemical component(s) potentially of toxicological significance not in the approved material?
If Yes: Sponsor responsible for safety assessment of modified material
If No: Proceed to next question
Does modification result in the removal of or reduction in, a chemical component(s) that contributes to the safety of the material?
If Yes: Sponsor responsible for safety assessment of modified material
If No: Proceed to next question
Does the material contain a component(s) present at ±10% compared to the generally expected composition of the approved material, where the component is of known therapeutic activity?
If Yes: Data sufficient to justify safety-in-use of modified material?
If No: Apply to TGA for evaluation
If Yes: Self assessment complete. Retain documentation for possible TGA audit
If No: Proceed to next question
Does the material contain a component(s) present at ±20% compared to the generally expected composition of the approved material, where the component is unknown or not associated with its therapeutic activity?
If Yes: Data sufficient to justify safety-in-use of modified material?
If No: Apply to TGA for evaluation
If Yes: Self assessment complete. Retain documentation for possible TGA audit
If No: Ingredient may be used in medicines
Glossary
| Term | Definition |
|---|---|
| Herbal material | Means a plant or part of a plant (defined by its botanical scientific name according to the binomial nomenclature system and by the plant part), whether whole, fragmented, cut or ground, and in an unprocessed state (whether fresh or dried). |
| Therapeutically active | Components that are chemically defined substances, or groups of substances, which in an isolated state exert the same or similar therapeutic effects as the total extract. These include sennosides (Senna), silymarin (Silybum) and kavalactones (Kava). |
| Pharmacologically relevant | Components that are chemically defined substances, or groups of substances which, in an isolated state, do not exert the same therapeutic effect as the total extract, but which are accepted to contribute to the therapeutic activity of the herbal preparation. These include hypericin (Hypericum) and flavonoids (Crataegus). |
| Unprocessed herbal material | Is the same as "herbal material", which means a plant or part of a plant (defined by its botanical scientific name according to the binomial nomenclature system and by the plant part), whether whole, fragmented, cut or ground, and in an unprocessed state (whether fresh or dried). |
Footnotes
- This is the amount of a component that falls within the typical range based on batch analysis, expected natural variation, historical or other data.
- The absence of a particular ingredient may have important consequence on safety and efficacy. For example, the absence of a low amount of a concomitant antioxidant may permit the formation of toxic oxidation products; or, the absence or low amounts of an oil may inhibit the absorption of lipophilic components.
- The component(s) ‘profile generally expected’ in the approved material will be a range, not a single value (point or target).
- To provide to the TGA. Fees and charges may apply to this evaluation.
Page history
Title changed from 'Guidance on the use of modified unprocessed herbal materials in complementary medicines' to 'Using modified unprocessed herbal materials in complementary medicines' as part of migration to new 'Guidance' content type:
- Consistent ‘Purpose’ heading.
- ‘Legislation’ section to clearly show which laws the Guidance relates to.
- ‘Page history’ section replaces document version history.
- New page navigation features.
- Updated page summaries.
- Complex images include long descriptions.
- New ‘Save as PDF’ feature.
Original publication
Title changed from 'Guidance on the use of modified unprocessed herbal materials in complementary medicines' to 'Using modified unprocessed herbal materials in complementary medicines' as part of migration to new 'Guidance' content type:
- Consistent ‘Purpose’ heading.
- ‘Legislation’ section to clearly show which laws the Guidance relates to.
- ‘Page history’ section replaces document version history.
- New page navigation features.
- Updated page summaries.
- Complex images include long descriptions.
- New ‘Save as PDF’ feature.
Original publication

