Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
If you are a software developer, manufacturer or product sponsor it is your responsibility to check whether your software or digital product is excluded from our regulation before you release it for supply in Australia.
There are currently 15 excluded software categories listed in Schedule 1 of the Therapeutic Goods (Excluded Goods) Determination 2018 (the Determination).
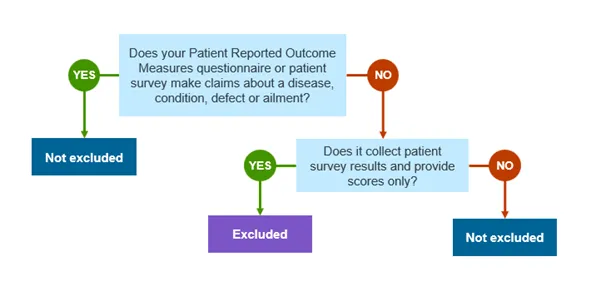
This guidance relates to software intended to be used as a patient reported outcome measures (PROMs) questionnaire or patient survey, which may be excluded from our regulation under item 14D of the Determination.