Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
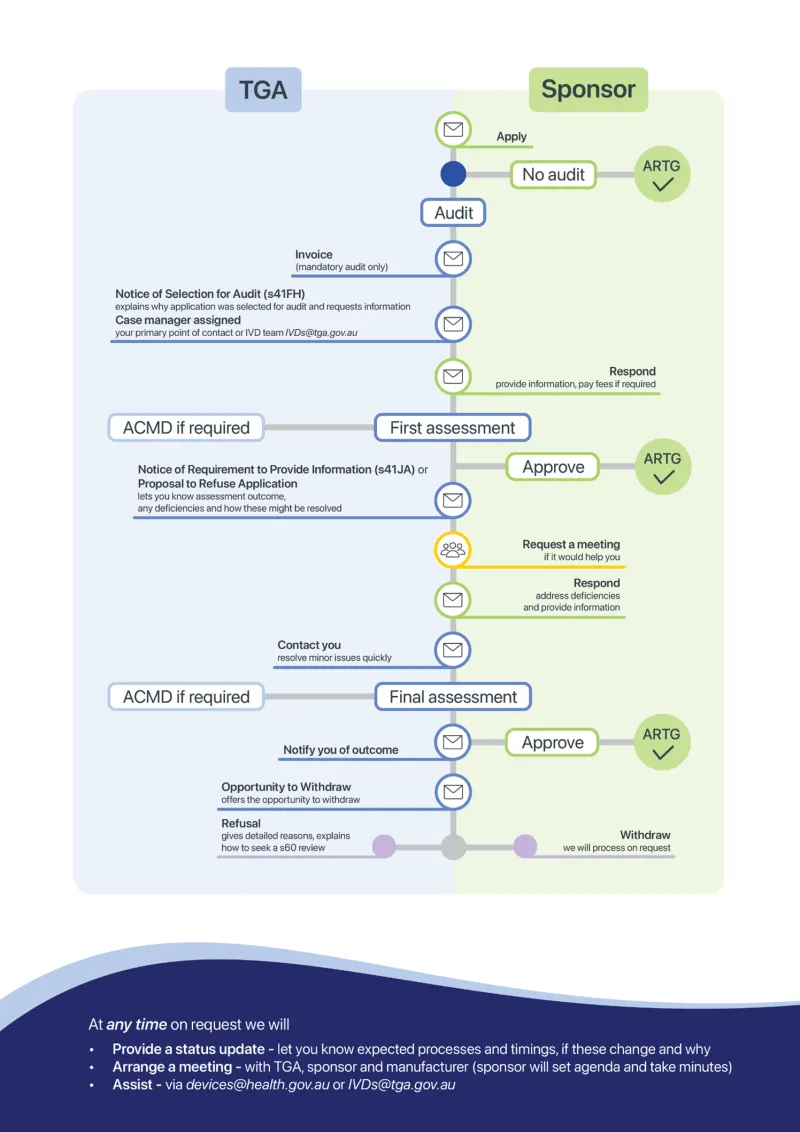
This guidance is for sponsors of applications for inclusion of medical devices, including in vitro diagnostic (IVD) devices, in the Australian Register of Therapeutic Goods (ARTG).
The application audit process verifies that medical devices meet legislative requirements for inclusion in the ARTG.
This document explains the case management of applications selected for audit and includes both mandatory and non-mandatory application audits.
This document does not apply to device change or variation applications.