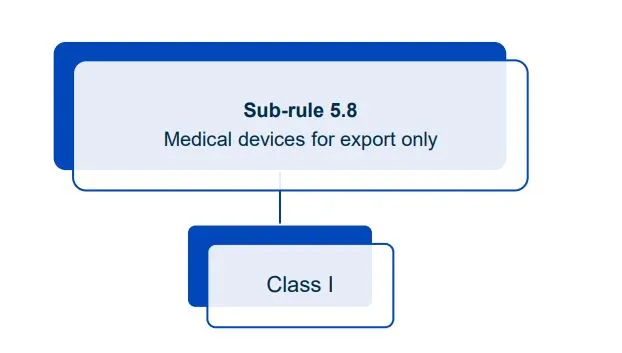
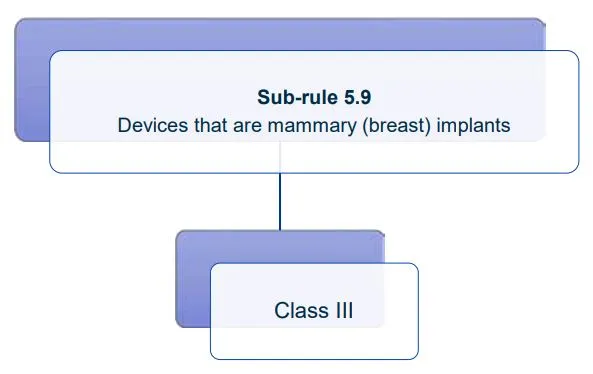
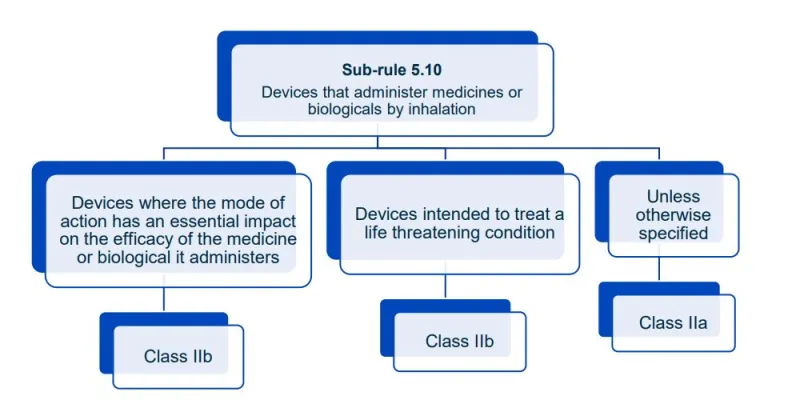
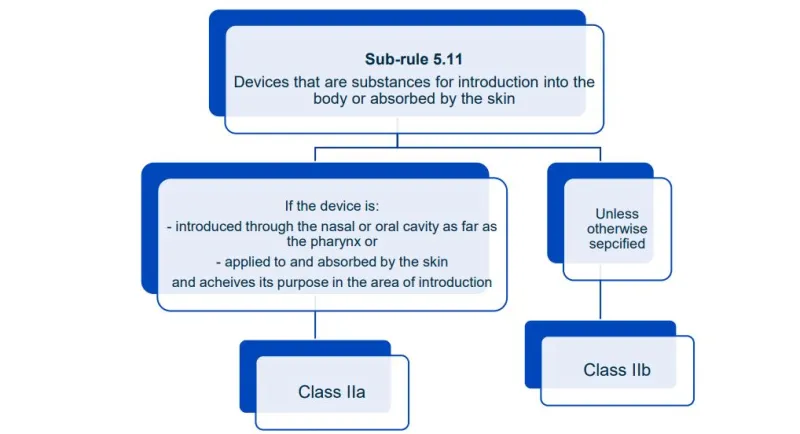
Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
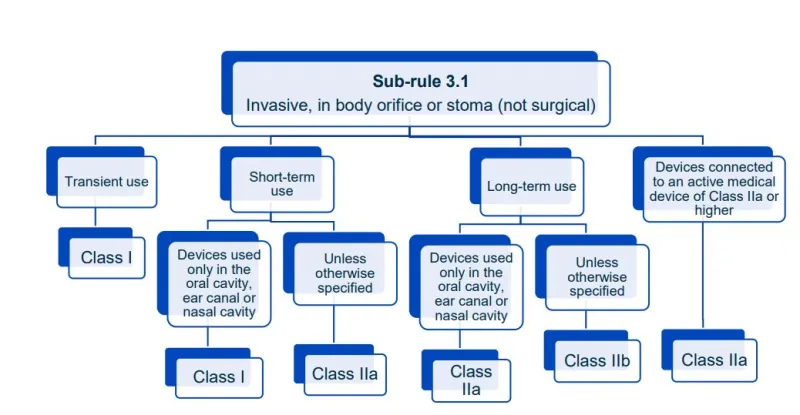
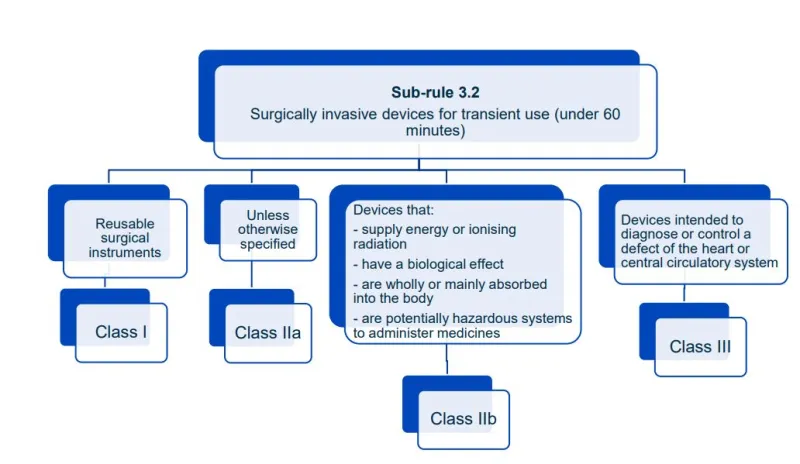
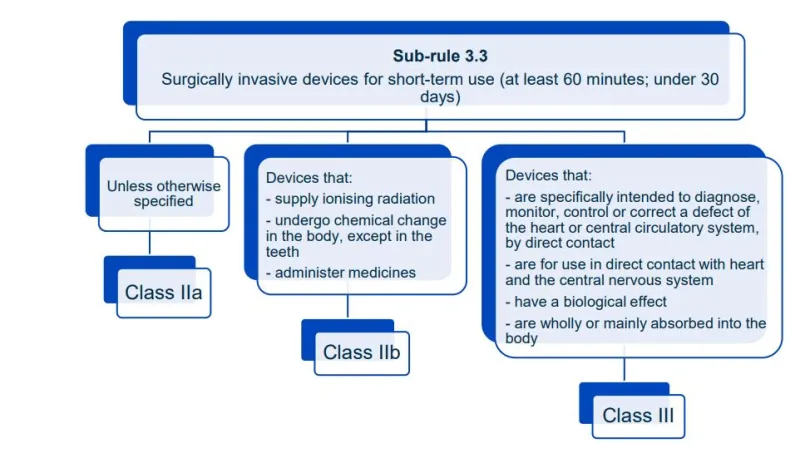
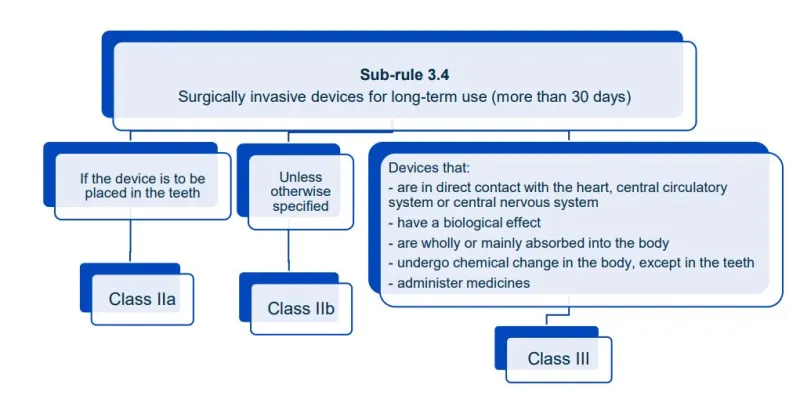
This document aims to help manufacturers in correctly classifying their devices. It is part of the Steps to supply for device manufacturers.
Read this guidance along with MD Regulations
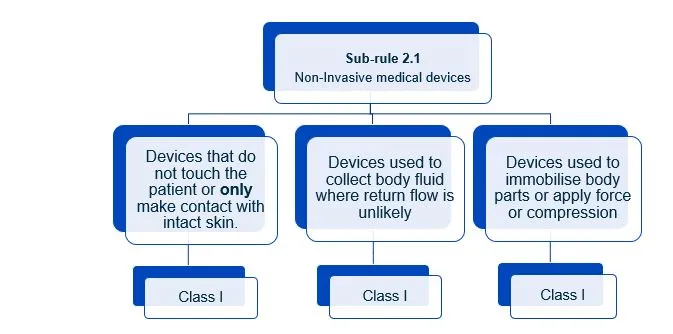
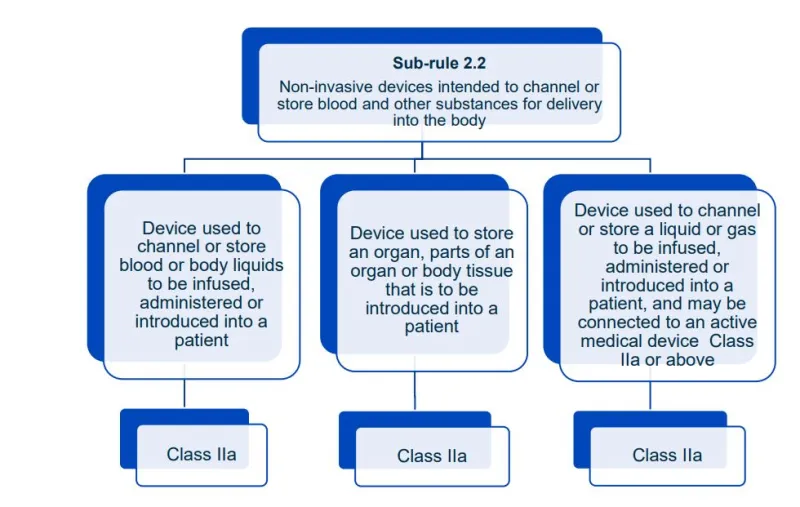
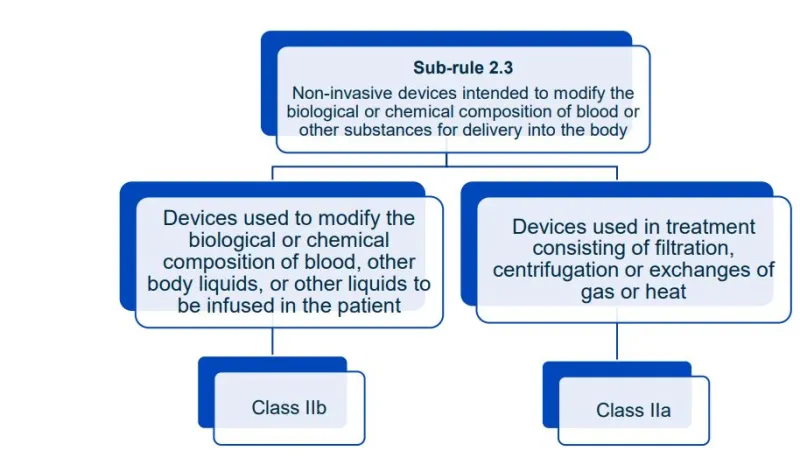
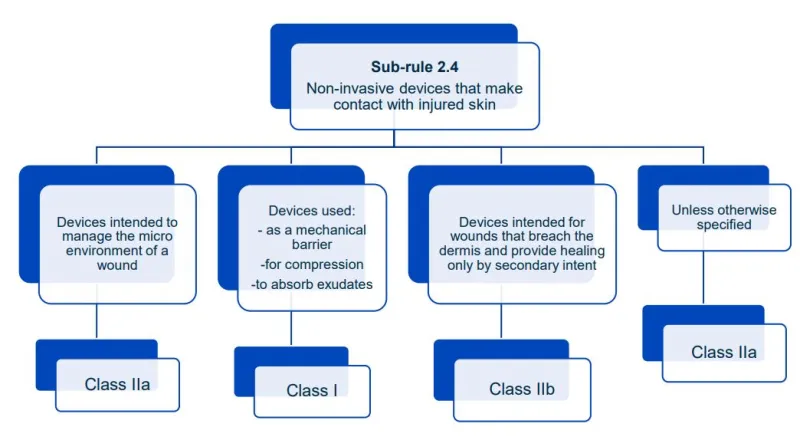
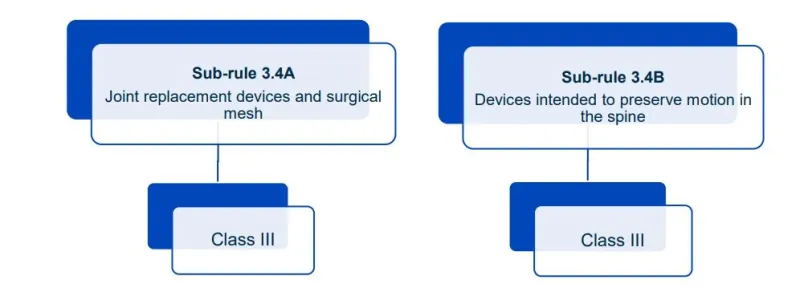
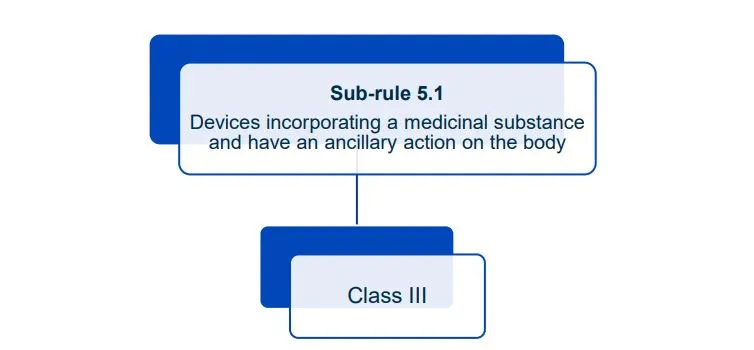
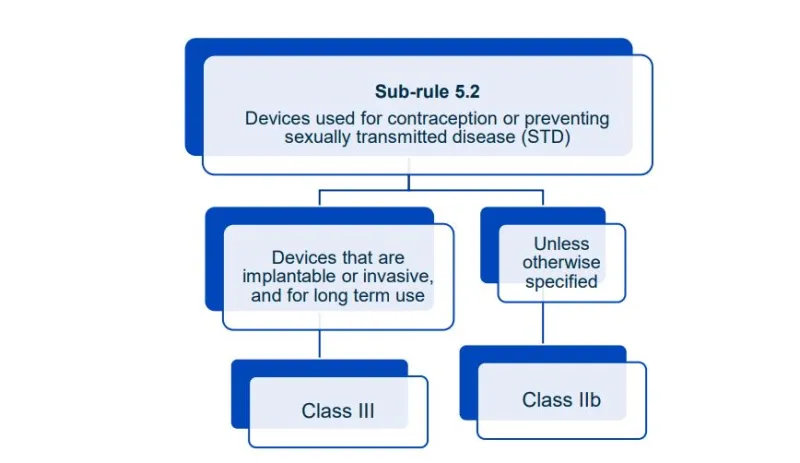
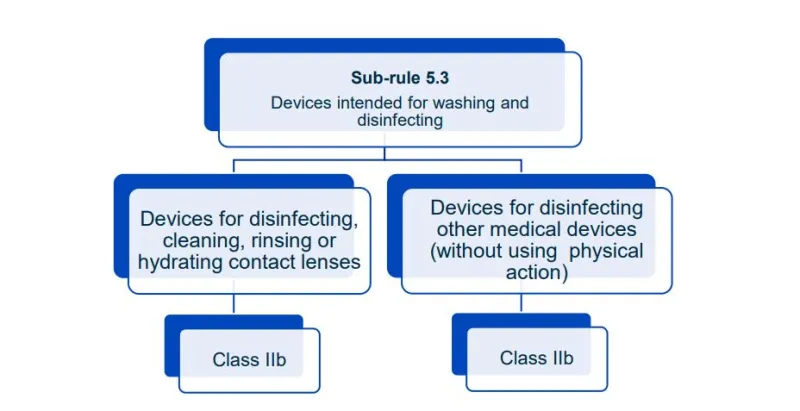
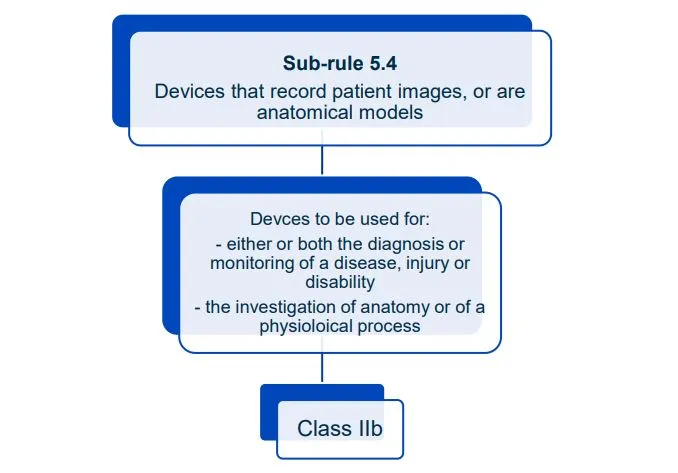
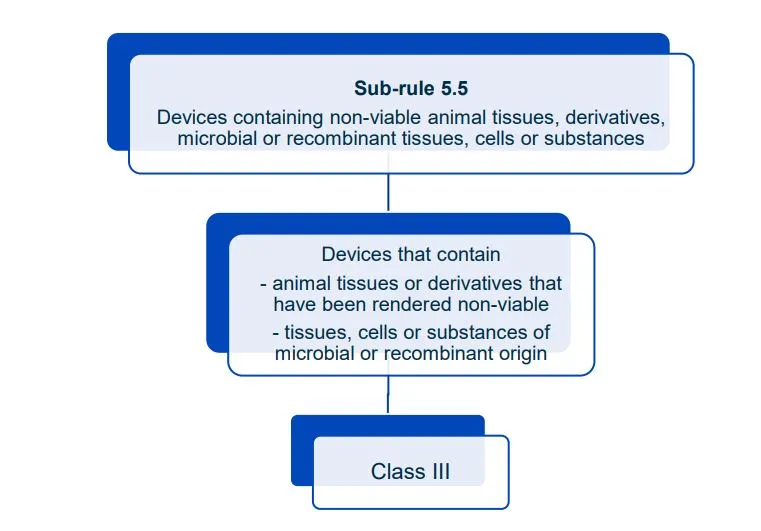
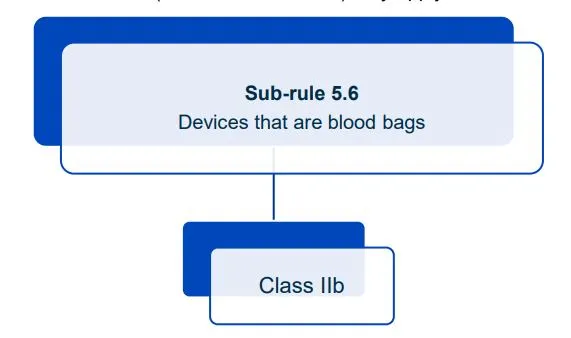
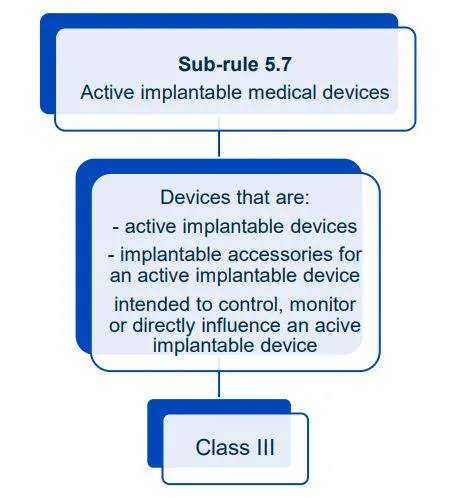
This document will help you understand medical device classification rules. It's not
a substitute for reading the Regulations and Explanatory Statements.
This document does not include guidance on:
- whether your product is a medical device. See Confirm your product needs to be in the ARTG as a medical device
- boundary and combination products. See Understanding rules for boundary and combination products
- classification of in vitro diagnostic (IVD) medical devices. See Classifying in-vitro diagnostic medical devices (IVDs) for supply in Australia
- medical devices intended to be supplied sterile
- medical devices with a measuring function.