Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
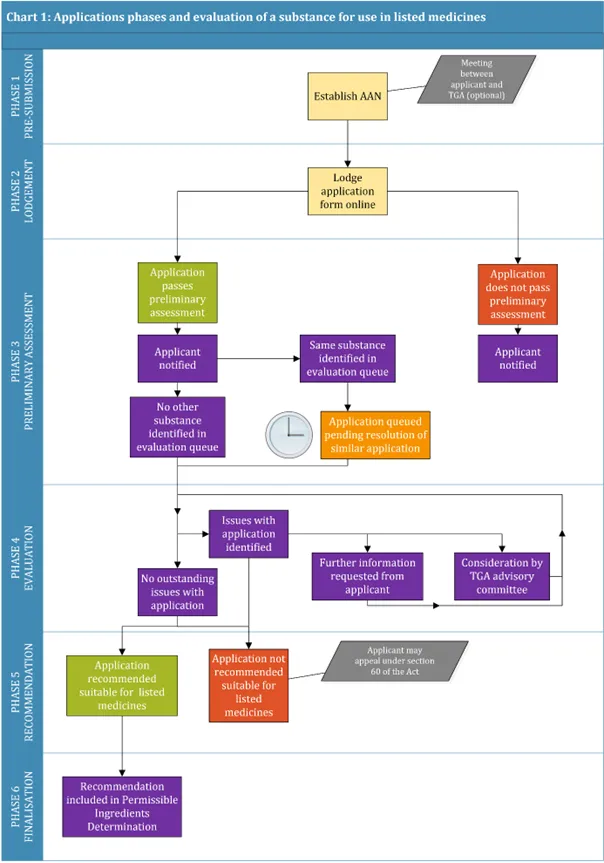
The Application requirements for new substances in listed medicines (ARNS), provides guidance for applicants requesting evaluation of a substance for use in listed medicines [AUST L listed medicines and AUST L(A) assessed listed medicines]. It replaces guidance formerly included in the ARGCM V 8.0 Part C.
The guidance includes:
- Application process for new substances
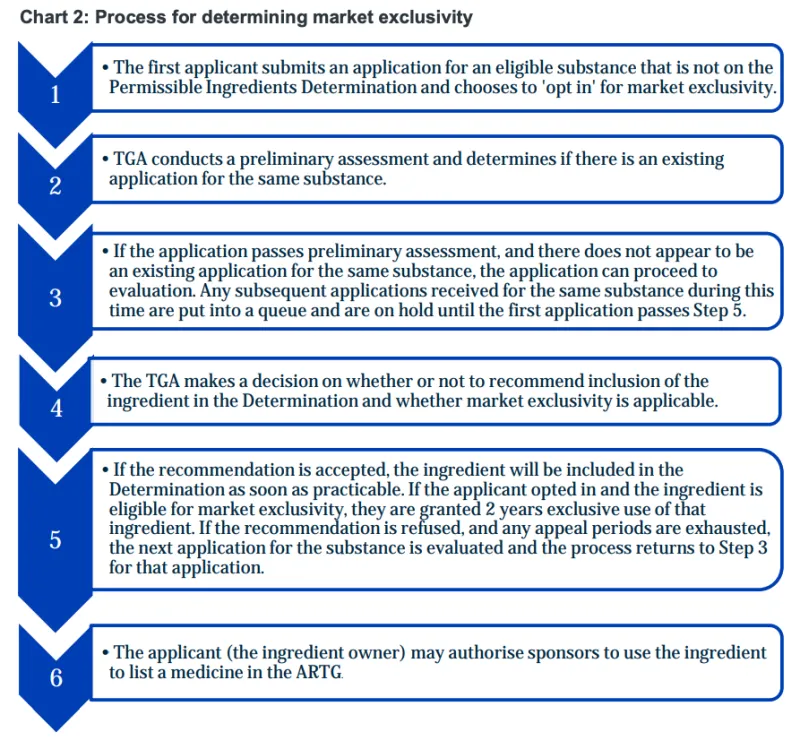
- How the TGA regulates market exclusivity of a new substance before and after approval
- Administrative, quality and safety information requirements for a substance application
- A summary of core information requirements for different substance types