Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
The Australian Government is strengthening patient safety by introducing Unique Device Identification (UDI) for medical devices.
UDI supports the identification of medical devices and other medical device reforms.
It is designed to improve the effectiveness of the regulatory framework, including the management of post-market safety-related activities, such as recalls.
When fully implemented in healthcare, UDI can provide faster identification of devices implanted into patients in the event of an adverse event, safety alert or recall.
UDI can support identification and removal of those devices from storage and distribution to prevent further use.
It also allows patients, consumers, and health professionals to access product information in the Australian UDI Database (AusUDID) about the devices that they use.
The AusUDID provides an easy to access and consistent location for up-to-date product information.
UDI could improve device performance assessment by regulatory bodies, clinical quality registries and device manufacturers through accurate product identification that better supports comparative studies.
With the introduction of UDI, Australia joins a globally harmonised approach that can supports more accurate identification of medical devices.
How UDI works
UDI facilitates traceability through the introduction of a unique device identifier for each model of medical device.
The unique device identifier is a combination of numbers, letters and symbols that show:
- the UDI-Device Identifier (UDI-DI) which indicates the model of medical device
- the UDI-Production Identifier (UDI-PI) which provides the production specific information such as lot or batch number.
The unique device identifier is issued by TGA recognised organisations known as Issuing Agencies.
The Issuing Agencies are responsible for ensuring the UDIs issued are unique and in accordance with global standards.
Device labels and all applicable levels of packaging for applicable devices are required to have a unique device identifier.
The identifiers must be labelled in a way that can be found and read by both people and machines (like a barcode).
The UDI may also be marked directly onto the medical device when the device is intended to be reused on multiple patients, like a scalpel.
Sponsors or manufacturers submit the UDIs and related device data to the AusUDID.
Patients, consumers and healthcare can find the information about the medical device in the AusUDID, by searching using the UDI.
Benefits
Every year, millions of devices are used on or implanted in patients in public, private and day hospitals and clinics.
In some instances, the ability to accurately identify medical implants has been challenging when an issue with an implant has been identified.
UDI allows clear and unambiguous identification of medical devices and facilitates access to information about the device.
This allows better identification of medical devices within Australia, strengthening patient safety and supporting quicker responses to device safety issues.
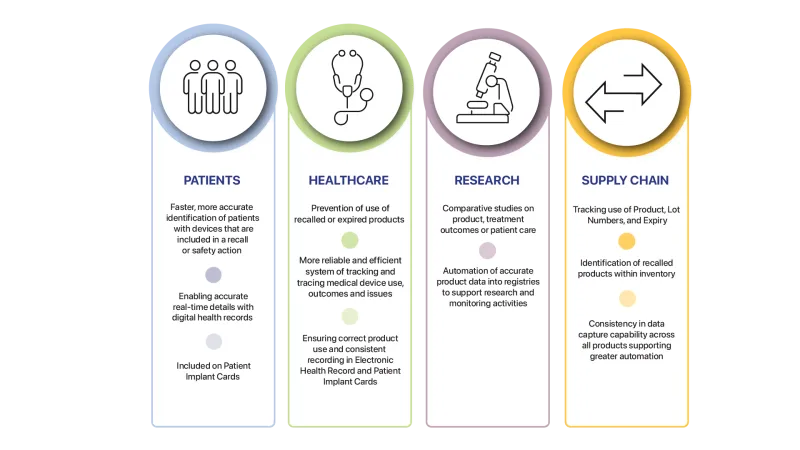
Further benefits made possible through the adoption of UDI are shown below: