GMP clearance Sponsor Information Dashboard (SID)
SID provides industry with regular updates about the backlog reduction, actual processing times, existing workloads, requesting prioritisation and other key messages for GMP clearance applications.
The backlog of Good Manufacturing Practice (GMP) Clearance Compliance Verification (CV) applications is continuing to reduce.
All backlog reduction strategies have been implemented and a recap has been published.
June update
We have updated the SID this month with two graphs. The first is re-introducing CV timeframe data but limiting this to applications submitted in the past two years. The second is a graph showing annual volumes of received and completed CV applications over the past 4 years.
We welcome any feedback you may have on the SID. Please send this to GMPclearance@health.gov.au.
We have also created links to newly published information on requesting GMP pre-submission meetings under ‘related resources’. This is a new initiative that has been successfully piloted with industry members during the past year.
The below data is from 1 June 2026 and will be updated every 4 to 6 weeks to ensure it remains current.
Backlog reduction progress
The graphs below illustrate the trend of GMP Clearance applications over time. Further details on actual numbers can be read from opening the drop-down menu under each graph.
Figure 1 shows the trend of the total number of CV applications in our lodgement queue over the past 12 months. The total number of CV applications steadily reduced in 2025, with a steep drop at the beginning of Q3 2025 due to the backlog reduction strategy. Since the backlog reduction strategy was introduced on 1 July 2025, the rate of reduction has also increased as expected. Since the end of Q2 2025, the total number of applications reduced from 2185 applications to 1203 to date, which equates to a reduction of ~44%.
Figure 1 - Total number of CV applications pending assessment between 1 March 2025 and 1 April 2026
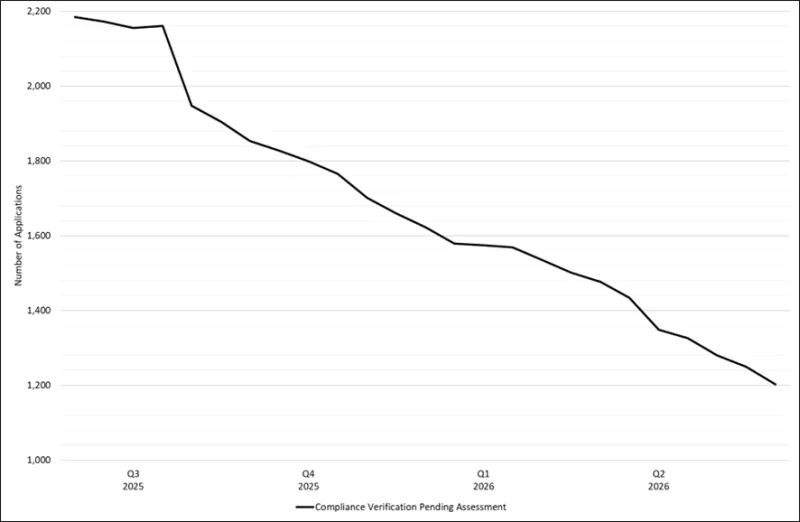
Figure 1 shows a line graph detailing the total number of Good Manufacturing Practice (GMP) Compliance Verification (CV) clearance applications between end of Q2 2025 and Q2 2026.
The vertical axis represents the total number of GMP clearance applications within the TGA’s application lodgement system.
The horizontal axis represents the date broken down by quarter and year.
The graph starts at 2185 applications in Q2 2025 and ends at 1203 applications in Q2 2026, illustrating a steady decrease in the backlog over the year.
Figure 2 shows the trend of applications in our CV streams over the past year with each line representing a different stream.
Both the Non-Sterile Active Pharmaceutical Ingredient (NS-API) and Non-Sterile Finished Product (NS-FP) streams have significantly reduced over the past year. More recently, the rate of reduction in the NS FP stream has increased following the allocation of additional resources, whilst the NS API stream has levelled off. Both sterile streams have remained relatively steady over the past year however recently; the Sterile Finished Product (ST FP) stream has started to reduce.
Figure 2 - number of CV applications in lodgement by stream between 1 June 2025 and 1 June 2026
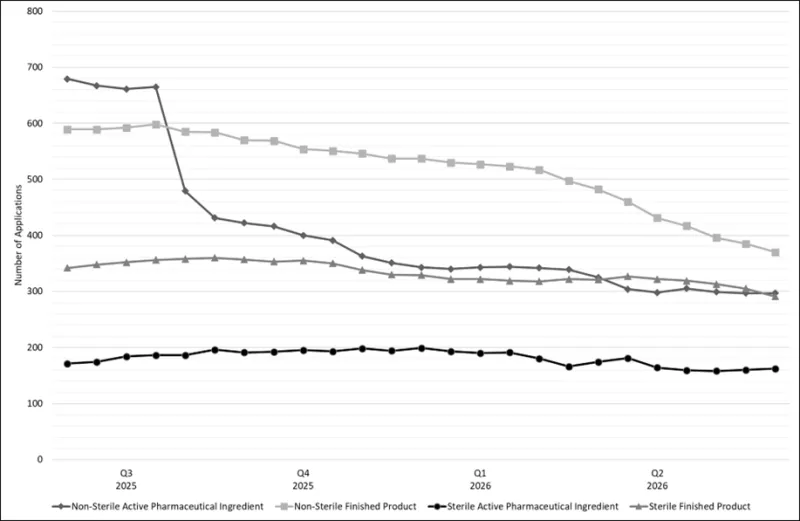
Figure 2 shows a line graph detailing the number of Good Manufacturing Practice (GMP) clearance compliance verification applications pending assessment in each stream between Q2 2025 and Q2 2026.
The vertical axis represents the total number of GMP clearance applications within the TGA’s application lodgement system.
The horizontal axis represents the date broken down by quarter and year.
There are four lines represented within the graph.
The line with a diamond marker at the top of the graph represents non-sterile active pharmaceutical ingredient compliance verification applications. This shows steady application numbers around ~680 up until the end of June 2025, followed by a sharp decrease to ~500 by August 2025. These numbers further decrease to ~297 by 1 June 2026.
The next line with a square marker represents non-sterile finished product compliance verification applications. The line shows a consistent decrease in application numbers from ~590 in Q2 2025 to ~370 by 1 June 2026.
The third line with a triangle marker represents sterile finished product compliance verification applications. This shows that there was a slight increase in application numbers from ~350 to ~380 between Q2 2025 and Q3 2025 but have decreased to ~291 by 1 June 2026.
The bottom line with a circle marker represents sterile active pharmaceutical ingredient compliance verification applications. This shows that application numbers have remained relatively steady between ~170 and ~200 within the last year.
Workload volumes
Figure 3 shows a snapshot of all GMP Clearance applications in the system at different stages of evaluation or queue categories. The largest number remains 'complete' applications in lodgement queues ready for evaluation (This includes both MRA and CV application types). The total number of all applications in the system on 1 June 2026 was 1286. This a reduction of 274 since the April update.
Figure 3 - total number of GMP clearance applications and the applications status as of 1 June 2026
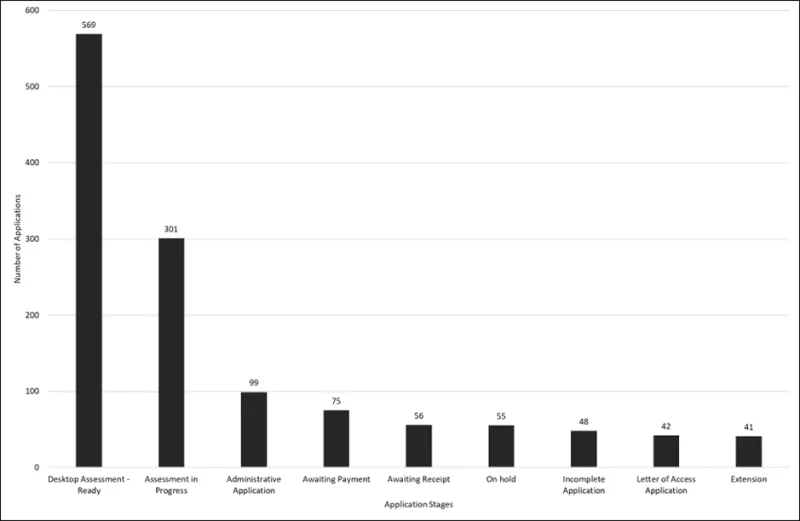
Figure 3 shows a bar graph detailing total numbers of Good Manufacturing Practice (GMP) clearance applications and the applications status.
The vertical axis represents the number of applications.
The horizontal axis represents the status of the applications.
There are nine bars:
The first bar from the left side of the graph represents complete applications awaiting assessment, there are 569 applications represented. This is reduced from 798 in April, with more applications being picked up for evaluation.
The second bar represents assessments in progress, that is applications currently being considered by evaluators, there are 301 applications represented. This has increased from 237 in April.
The third bar represents administrative applications awaiting assessment, this includes updates for manufacturer names and addresses as well as other application types, there are 99 applications represented, a decrease by 35 applications since April.
The fourth bar represents applications awaiting payment, there are 75 applications represented, a decrease of 1 since April.
The fifth bar represents applications awaiting receipt (screening), there are 56 applications represented, a decrease of 1 since April.
The sixth bar represents applications which are currently on hold (typically for GMP non-compliance investigations), there are 55 applications represented, an increase of 7 since April.
The seventh bar represents incomplete applications. There are 48 applications represented, decreasing by 23 since April.
The eighth bar represents letter of access applications, there are 42 applications represented, a decrease of 12 since April.
The ninth bar on the far right of the graph represents extensions. There are 41 applications represented as compared to 85 in April, which is a significant decrease of 44 since April.
CV processing timeframes (Rolling 2-year data)
Figure 4 shows the percentile ranges for application processing timeframes for applications submitted in the last two years for each CV stream, measured in TGA working days.
This graph has been re-introduced but now excludes data from application submitted more than two years ago. This gives Industry a more realistic snapshot of expected processing timeframes for future applications.
Each bar is divided into percentiles, which represent the percentage of applications completed within a certain number of days. For example, the 50th percentile means half of the applications were completed in that time or less. The percentiles build on each other - for example, the 50% mark includes all applications completed within the 10% and 25% ranges as well.
It is expected these percentiles will fluctuate as we return to a steady state and continue to manage requests for prioritisation.
Figure 4 - percentage of CV applications submitted and completed within the past two years as of 1 June 2026
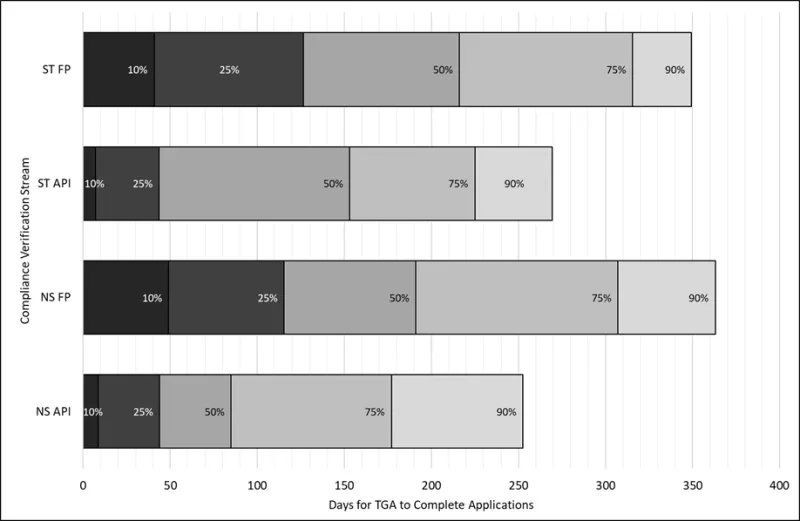
ST FP: sterile finished product applications
ST API: sterile active pharmaceutical ingredient applications
NS FP: non-sterile finished product applications
NS API: non-sterile active pharmaceutical ingredient applications
Figure 4 shows a bar graph detailing percentages of Good Manufacturing Practice (GMP) clearance compliance verification applications completed within specified TGA working days during the past two years.
The vertical axis represents the compliance verification application streams.
The horizontal axis represents the number of days taken by the TGA to complete applications.
There are four bars, the first bar from the bottom of the graph represents non-sterile active pharmaceutical ingredient compliance verification applications. This shows that:
- ten percent of applications are completed within 9 days,
- twenty five percent of applications are completed within 44 days,
- fifty percent of applications are completed within 85 days,
- seventy five percent of applications are completed within 177 days
- ninety percent of applications are completed within 253 days.
The second bar represents non-sterile finished product compliance verification applications. This shows that:
- ten percent of applications are completed within 49 days
- twenty five percent of applications are completed within 116 days
- fifty percent of applications are completed within 191 days
- seventy five percent of applications are completed within 307 days
- ninety percent of applications are completed within 363 days
The third bar represents sterile active pharmaceutical ingredient compliance verification applications. This shows that:
- ten percent of applications are completed within 7 day
- twenty five percent of applications are completed within 44 days
- fifty percent of applications are completed within 153 days
- seventy five percent of applications are completed within 225 days
- ninety percent of applications are completed within 270 days
The fourth bar represents sterile finished product compliance verification applications. This shows that:
- ten percent of applications are completed within 41 days
- twenty five percent of applications are completed within 127 days
- fifty percent of applications are completed within 216 days
- seventy five percent of applications are completed within 316 days
- ninety percent of applications are completed within 349 days
MRA processing timeframes (rolling 3 monthly data)
The figures below provide an overview of the target and actual processing times for certain application types, measured in TGA working days. The actual processing time applies for 90 percent of applications completed within the last 3 months.
-
30
Target MRA application processing time
-
26
Actual MRA application processing time
MRA applications have been consistently processed within our target processing time. With more evaluation resources focussed on the CV streams, we have been using the buffer previously built up in the MRA space. The actual processing time has therefore increased to 26 days. An increase of 6 days since April.
CV application annual volumes
Figure 5 shows a snapshot of GMP Clearance CV applications received and completed within the last four years since 2023. Overall, there is a general decline in applications received over time. In 2025, we completed 1704 CV applications, which is 57% more than received that year as we implemented the backlog reduction strategy and had more evaluators signed off for CV applications. The total number of applications completed year to date as of 1 June 2026 is 697.
Figure 5 - total number of GMP clearance CV applications received and completed per year as of 1 June 2026
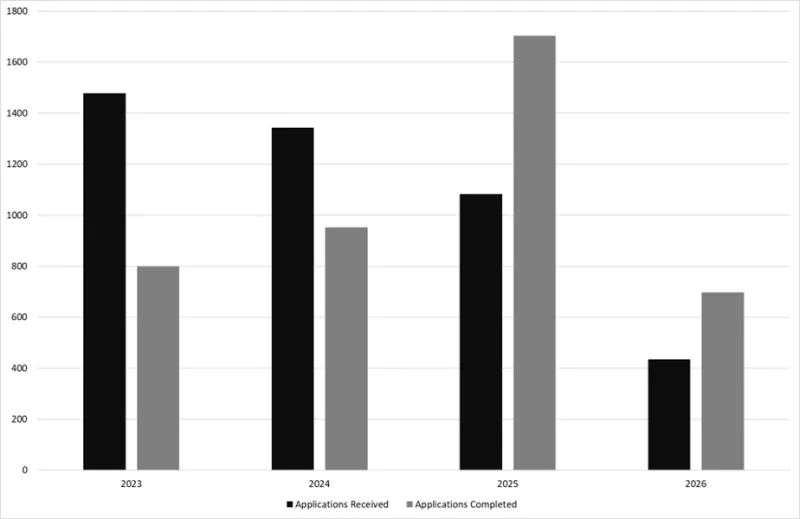
Figure 5 shows a clustered column bar graph comparing the number of Good Manufacturing Practice (GMP) clearance compliance verification applications received and completed across four years (2023-2026).
The vertical axis represents the number of applications.
The horizontal axis represents the calendar year.
There are four pairs of bars, with each pair representing a single year. Within each pair:
- the black bar represents applications received, and
- the grey bar represents applications completed.
The first pair of bars (2023) shows that:
- approximately 1,450 applications were received, and
- approximately 800 applications were completed.
The second pair of bars (2024) shows that:
- approximately 1,350 applications were received, and
- approximately 950 applications were completed.
The third pair of bars (2025) shows that:
- approximately 1,100 applications were received, and
- approximately 1,700 applications were completed.
The fourth pair of bars (2026) shows that:
- approximately 450 applications were received, and
- approximately 700 applications were completed.
Overall, the graph shows a decreasing trend in applications received over time, while applications completed increase significantly in 2025 with the implementation of backlog reduction strategies.
Application Quality (rolling 6-monthly data)
The figures below provide an overview of how many applications required a Request For Information (RFI) or were incomplete at the time of submission.
-
12 % of 346 applications
Percentage of Mutual Recognition Agreement (MRA) applications requiring an RFI
-
31 % of 799 applications
Percentage of incomplete CV applications
-
69 % of 799 applications
Percentage of Compliance Verification (CV) applications requiring an RFI
-
33 % of 58 applications
Percentage of TGA certificate applications requiring an RFI
-
73 % of 48 applications
Percentage of applications using a letter of access (LoA) requiring an RFI
The above data tells us that approximately 1 in every 3 CV applications is submitted incomplete. This adds pressure onto TGA staff and causes significant delays in processing times. We encourage sponsors to work with their manufacturers to ensure all required evidence is provided at the point of submission where possible.
Prioritisation requests
We will continue to process effective applications as quickly as possible. Medicine shortages remain a high priority for us, and we will continue to process these when sponsors have provided the required information.
Please note that we will liaise internally with the medicine shortages team to verify requests and seek additional information such as supply and market data.
For any request for prioritisation, you must provide the required information outlined below via email:
- The product name and existing ARTG number (where applicable)
- The category of medicine (Complementary, Non-prescription, Prescription etc.)
- The product submission type and submission number and any applicable milestone dates (for example, priority review pathway or NCE, PM-XXXX-XXXXX-X-X etc.)
- For variations, you need to provide information about the change as certain changes made by manufacturers may require additional GMP evaluation.
- For requests linked to reportable medicines, the medicines shortage notification number (for example MS-XXXX-XX-XXXXX-X)
- Any other information to help us understand the urgency of the situation. For example, existing stock levels or run-out dates, negative effect on patients or business etc.
Featured notices
-
News articlesA review of the GMP Clearance backlog reduction strategies that have been implemented and ongoing monitoring and reporting communication.
-
News articlesThe GMP Clearance SID now contains additional data as well as progress information on the Compliance Verification (CV) backlog reduction strategy.
-
News articlesInformation on the completion of automatic extensions for Good Manufacturing Practice (GMP) clearances and next steps.
Related resources
-
FormsThis form is for applicants of medicines and biologicals to request for a GMP pre-submission meeting.
-
PageFind out about obtaining Good Manufacturing Practice (GMP) clearance for an overseas manufacturing site.
-
User guideThis user guide is for sponsors wanting to submit GMP clearance application by the TGA Business services (TBS) portal.
-
User guideCheck out our user guide to assist sponsors when using the code tables for GMP Clearance applications.

