Consent to import, supply or export therapeutic goods that do not comply with standards - information for industry
Information on requirements and process to apply to import, supply or export therapeutic goods that do not comply with standards.
Overview
Medicines and other therapeutic goods must comply with applicable standards to be supplied in Australia.
It is an offence to import, supply or export therapeutic goods that do not comply with a standard applicable to the goods, unless the prior consent in writing of the Secretary has been given (see section 14 the Therapeutic Goods Act 1989) or a civil penalty may be payable (see section 14A of the Act).
Sections 14 and 14A apply to therapeutic goods that are registered or listed in the Australian Register of Therapeutic Goods (ARTG) under Part 3-2 of the Act including:
- registered and listed medicines,
- registered and listed therapeutic devices, and
- export only listed therapeutic goods,
and also to therapeutic goods for which standards under section 10 apply but are exempt from registration or listing under Part 3-2 such as:
- medicines that are blood or blood components manufactured by the holder of a licence to manufacture blood and blood components and
- medicines that are exempt from registration or listing because they are extemporaneously compounded for a particular person for therapeutic application to that person, or have otherwise been approved under Part 3-2 (such as under section 19A of the Act).
The provisions also apply in relation to the import, supply and export of biologicals that are included in ARTG under Part 3-2A of the Act and to biologicals that are exempt from being so included or otherwise approved under that Part. However the provisions do not apply to a biological if it meets the prescribed circumstances as set out in regulation 33A of the Therapeutic Goods Regulations (exceptional release).
These provisions do not apply to medical devices. For more information, or to apply for consent to import, supply or export a medical device that does not comply with the Essential Principles, go to Essential Principles - consent for noncompliance.
What is a standard?
A standard for this purpose is any of the following:
- a standard that is constituted by the matters in an order made by the Minister under section 10 of the Act;
- if the goods are the subject of one or more monographs in the British Pharmacopoeia - a standard that is constituted by the statements in those monographs, as interpreted in accordance with the General Notices section of the British Pharmacopoeia;
- if the goods are the subject of one or more monographs in the European Pharmacopoeia - a standard that is constituted by the statements in those monographs, as interpreted in accordance with the General Notices section of the European Pharmacopoeia;
- if the goods are the subject of one or more monographs in the United States Pharmacopeia National Formulary - a standard that is constituted by the statements in those monographs, as interpreted in accordance with the General Notices section of the United States Pharmacopeia National Formulary.
Standards that are specified in an order under section 10 of the Act include (but are not limited to) the following (these are the ones most likely to be the subject of an application):
- Therapeutic Goods Order No. 91 - Standard for labels of prescription and related medicines
- Therapeutic Goods Order No. 92 - Standard for labels of non-prescription medicines
- Therapeutic Goods Order No. 100 - Therapeutic Goods (Microbiological Standards for Medicines) Order 2018
- Therapeutic Goods Order No. 101 - Therapeutic Goods (Standard for tablets capsules and pills) Order 2019
- Therapeutic Goods Order No. 102 - Therapeutic Goods (Standard for Blood and Blood Components) Order 2019
- Therapeutic Goods Order No. 107 - Therapeutic Goods Order (Standard for biologicals - Labelling requirements) Order 2021
- Therapeutic Goods Order No. 108 - Therapeutic Goods (Standard for human cell and tissue products - Donor screening requirements) Order 2021
- Therapeutic Goods Order No. 110 - Therapeutic Goods (Standard for Therapeutic Vaping Goods) Order 2021)
How to obtain consent
Any person in relation to whom:
- a medicine or other therapeutic good is in the ARTG under Part 3-2 of the Act, or
- a biological is in the ARTG under Part 3-2A of the Act,
can make an application to the TGA.
An application can include goods in multiple ARTG entries provided:
- all the issues in relation to granting consent are the same for all the goods, and
- the non-compliance in relation to the goods relate to the same part or parts of a standard applicable to the goods.
For instance:
- multiple ARTG entries of goods that do not comply with paragraph 8(1)(g) of TGO 91/92 (the expiry date preceded by the expiry date prefix) could be included in one application.
Those wanting to import and/or supply or export a product that is exempt or otherwise approved under Part 3-2 or Part 3-2A that does not comply with a standard that applies to the product should contact the TGA.
Fees
An application will not be considered unless the relevant processing fee has been paid. The fee is payable in relation to each application (see item 1A, Part 1 of Schedule 9 of the Therapeutic Goods Regulations 1990). This means one fee can cover multiple ARTG entries in the circumstances described above.
From 1 July 2020, there will be a fee for processing of applications for consent for biologicals.
What information must be provided?
The applicant has the responsibility of satisfying the TGA that consent should be given. The applicant should provide information:
- identifying the relevant part of part (or parts) of the standard that is not complied with;
- the relevant products (including batch numbers where relevant);
- the circumstances giving rise to the non-compliance;
- the risks associated with the non-compliance and steps being taken to address any such risks (for instance, a proposed letter to healthcare professionals);
- if there is potential for supply shortage of compliant goods if consent is not granted, an indication of the impact on immediate and future supply, including where relevant
- how long the non-compliance is likely to last (i.e. what period/batches is the consent to apply for/to);
- strategies to rectify the non-compliance and expected timeframe for implementation (where relevant).
The TGA may request additional information in order to come to a view about an application.
It is a serious offence to give false or misleading information to the TGA for the purposes of making an application for consent under section 14/section 14A (see sections 136 and 137 of the Commonwealth Criminal Code).
What does the TGA decision-maker take into account?
Ongoing compliance with applicable standards is an important factor in ensuring that the therapeutic goods supplied to the Australian public are of the appropriate quality, safety and efficacy.
Consent to not complying with an applicable standard will not be given unless the TGA is satisfied that, whether because of the imposition of conditions or otherwise, the safety, quality, safety and efficacy and/or the safe and effective use of the product will not be adversely affected.
The factors that the TGA delegate can take into account for that purpose include:
- clinical need for an uninterrupted supply and extent to which suitable alternatives are available;
- the extent to which the imposition of appropriate conditions will sufficiently mitigate any risks;
- whether it is appropriate to limit the consent by reference to time and or number of batches;
- whether the issue giving rise to the lack of compliance was foreseeable or avoidable.
Duration of consent
The consent can be provided in relation to specific batches and/or be limited in duration. Consent is generally not granted for a period longer than 2 years. In exceptional circumstances, consent can be granted for a longer period. In all circumstances the proposed duration must be justified in the application.
Imposition of conditions
Delegates may impose any conditions under section 15 of the Act to mitigate any risk and to ensure that the safety, quality and efficacy or the safe and effective use of the product will not be adversely affected. In determining conditions, consideration is also given to what is required to ensure relevant information is provided about the safe use of the product down the supply chain.
It is an offence to breach a condition imposed on a consent (see section 15 of the Act) and civil penalties are payable for such a breach (see section 15AA of the Act).
Publication of consent decisions
The TGA is required to publish particulars of any consent given in the Commonwealth Gazette or on the TGA's website. In the past, the consents have been published in the Gazette but from 1 February 2016 these have been published on the website in a form that is searchable. They will remain accessible on the website.
Where the decision is to reject, the TGA must notify the sponsor within 28 days and give reasons. The TGA is not required to publish these decisions.
Rights of review
The Secretary must notify the applicant of any refusal of consent within 28 days after making the decision and provide reasons. The applicant has a right under section 60 of the Therapeutic Goods Act 1989 to seek internal review of such a decision within 90 day of notice of the decision being given to the person. The applicant can also seek review by the Administrative Appeals Tribunal of such an internal review.
The applicant also has a right to seek internal review under section 60 of the Act of any decision to impose conditions (under section 15 of the Act) on a consent within 90 days of being informed about the decision. The applicant can also seek review by the Administrative Appeals Tribunal of such an internal review.
Details about rights of review are included in the notices of decisions provided to applicants.
Reporting of medicine shortages
Sponsors may request priority review of a section 14 consent to mitigate a shortage of a reportable medicine after submitting a medicine shortage notification.
Market action and section 14 consent processes
The Procedure for recalls, product alerts and product corrections (PRAC) market action processes and the section 14/14A consent processes usually occur at different stages in the supply chain, as illustrated in the following graphic.
Market action and section 14 consent processes
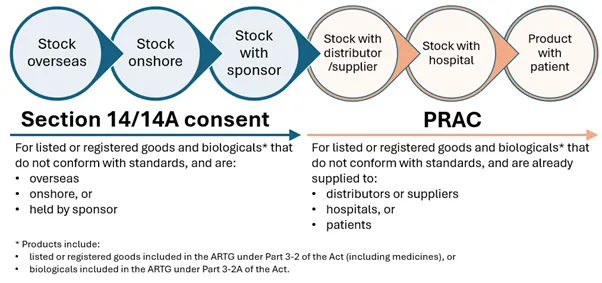
The image is a horizontal flow diagram illustrating pathways for handling non‑conforming therapeutic goods under Section 14/14A consent and PRAC.
Across the top are seven connected circular nodes, showing the movement of stock:
- Stock overseas
- Stock onshore
- Stock with sponsor
- Stock with distributor/supplier
- Stock with hospital
- Product with patient
The first three circles (overseas, onshore, sponsor) are grouped together as Section 4/14A consent. The next three (distributor/supplier, hospital, patient) are grouped together as PRAC.
This visually separates the process into two regulatory phases.
Under 'Section 14/14A consent', text explains:
For listed or registered goods and biologicals* that do not conform with standards, and are:
- overseas
- onshore, or
- held by sponsor
Under 'PRAC', text explains:
For listed or registered goods and biologicals* that do not conform with standards, and are already supplied to:
- distributors or suppliers
- hospitals, or
- patients
An asterisked note clarifies that products include:
- listed or registered goods included in the ARTG under Part 3‑2 of the Act (including medicines), or
- biologicals included in the ARTG under Part 3‑2A of the Act.
However, there are some circumstances where both processes may need to be applied concurrently. For example:
- a medicine defect is found after supply has occurred - requiring a market action (product alert or product correction), and
- at the same time, the same sponsor needs to maintain supply despite the goods not complying with an applicable standard - requiring a section 14/14A consent application for any remaining stock in their possession or for future batches.
Both processes aim to protect patient safety while maintaining continuity of supply. Sponsors must keep clear records and communicate with us throughout these processes.
Related links
Page history
Added section 'Market action and section 14 consent processes'.
Added section 'Market action and section 14 consent processes'.

