Class 4 in-house IVDs: using the online application and notification forms
Guidance to help sponsors fill out the online form in TGA Business Services to apply for TGA approval to supply a class 4 in-house IVD. Also, guidance for laboratories to notify the TGA about supply of exempt Class 4 in-house IVDs used to screen donors for manufacture of Faecal Microbiota Transplant (FMT) products.
Notify the TGA about exempt Class 4 in-house IVDs
Class 4 in-house IVDs used to screen donors for manufacture of FMT product are exempt from inclusion in the ARTG (Schedule 4, Part 2.10A of the Regulations). Eligible laboratories must notify the TGA about these exempt tests (Schedule 3, Part 6B.8 of the Conformity Assessment Procedures).
Laboratories should download, complete, and email the form below to: IVDs@tga.gov.au. There is no fee for submitting this notification form.
Apply for approval to supply a Class 4 in-house IVD
The rest of this guidance is for sponsors who are applying to include class 4 in-house IVDs on the ARTG.
TGA Business Services
The online application form is in TGA Business Services.
- Before your organisation uses TGA Business Services for the first time, you need to apply for a client identification number.
- For help with using TGA Business Services, including resetting your password, go to TGA Business Services - how to use this site.
Manufacturer evidence
Before you can apply for a Class 4 in-house IVD, you need to have submitted your manufacturer evidence to the TGA, and we need to have accepted it.
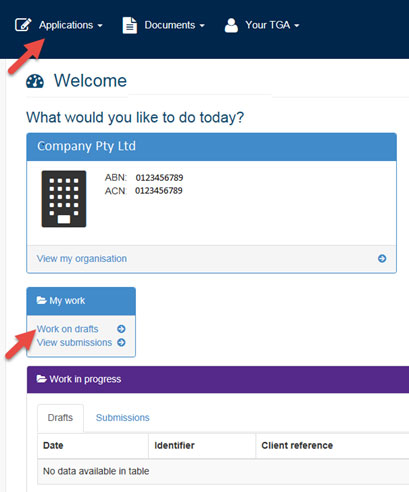
The dashboard
First, log in to TGA Business Services.
Across the top of the dashboard there are three main menus: Applications; Documents; and Your TGA. If you have the financial role, there is an additional Financials menu.
- To begin a new application, select the Applications menu.
If you want to open an existing draft form, select Work on drafts from the My work menu.
Starting a new Class 4 in-house IVD application
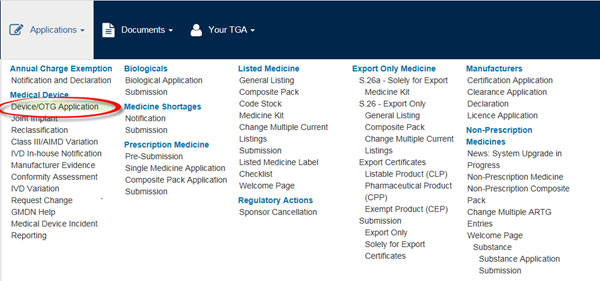
- Select Applications from the top menu. This will open a list of application types.
Select Device/OTG Application.
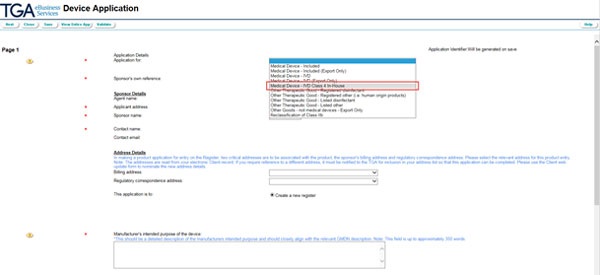
A blank Device application form is then displayed, starting with the Application page 1.
Note that Help texts are available throughout the form using the yellow '?' buttons on the left.
To change your device application into an IVD device application, select 'Medical Device - IVD Class 4 In-House' from the drop down menu at Application for.
Completing the form
At any stage you can save the application form to your drafts by clicking the Save button on the bottom of your screen.
Once you selected 'Medical Device - IVD Class 4 In-House', sponsor, address and application class details will auto-populate from your client details.
- Verify the auto-populated details and when necessary, change them using the drop down menus.
Provide a detailed description (up to 350 words) of the intended purpose of the device that closely aligns with the relevant GMDN description.
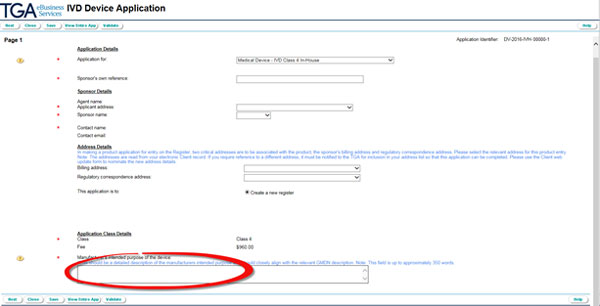
- Click the Next button at the bottom of the page.
Select the Manufacturer for Evidence from the drop down menu on page 2.
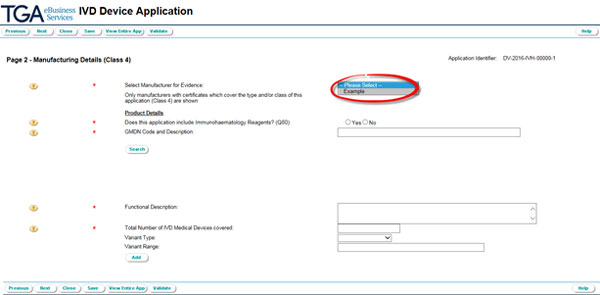
This drop down menu only shows manufacturers with accepted manufacturer's evidence that covers the type and class 'IVD class 4 in-house'. If the intended manufacturer is not shown, make sure that you have submitted the manufacturer's evidence, and that we have accepted it.
Select the Evidence number from the drop down menu that pops up after you have selected the manufacturer and indicate the authority that issued the evidence.
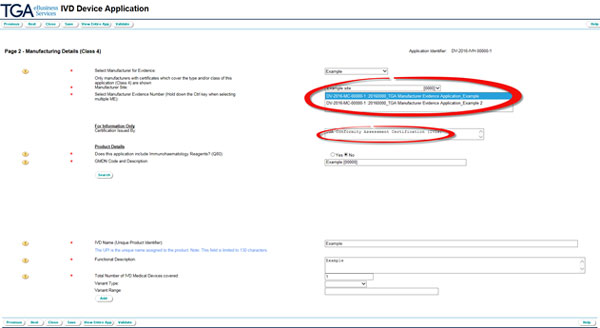
Indicate whether the device includes immunohaematology reagents.
If 'yes' is selected, the form will no longer display the IVD name (Unique Product Identifier), because this is not applicable to immunohaematology reagent IVD applications.
Use the search button to take you to the GMDN code search screen. Enter a search term and select the GMDN code for the application. Click the OK button.
Note: If the application is for an immunohaematology reagent, only GMDN codes for immunohaematology reagents will be displayed.
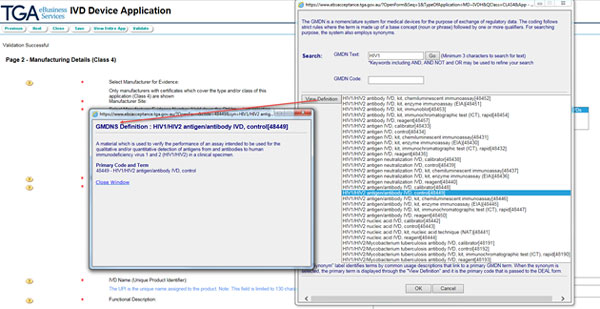
- If your IVD is not an immunohaematology reagent, enter the Unique Product Identifier. This text will be used as the 'product name' for the ARTG entry.
- For the Functional Description, enter a description of how the device achieves its intended purpose.
- For the total number of IVD medical device variants covered:
- Enter '1' if the IVD is not an immunohaematology reagent
- Specify the total number of variants that you are applying for, if the IVD is an immunohaematology reagent.
- Variant type is only applicable to immunohaematology reagents. For immunohaematology reagents:
- Select as the type 'Immunohaematology reagents'
- Provide details in the free text Variant range field. Click the Add button to confirm.
- Click the Next button at the bottom of the screen.
Submitting your application
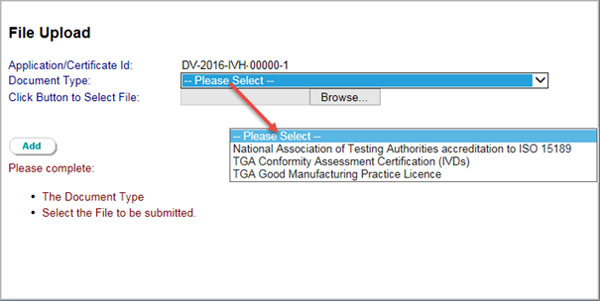
- Click the Add button on top of page 3 to upload supporting documentation.
Select the Document Type from the drop down menu and select Browse to search for files. Then, select the file to be submitted and click the Add button once more to confirm.
Read the Declaration and agree or decline.
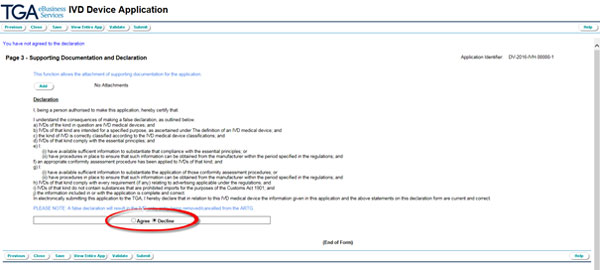
You have to agree to the declaration in order to submit your application.
Click the Validate button at the bottom of the screen to run a check whether all mandatory questions have been answered.
Once the validation is complete:
If you only have drafter rights, click the Save button at the bottom of the screen. Ask a person in your organisation that has submitter rights to verify the application and submit.
If you do not have the submitter role, there will be no Submit button at the bottom of your screen.
- Only someone with the submitter role can submit the application. Use the Submit button at the bottom of the screen.
- We will only process your application once we have received payment. When you submit the application, an invoice will be automatically generated and will be visible if you have the financial role. Please note, we will not send you a paper copy of the invoice by post.
Print version
Page history
Added the 'Exempt Class 4 in-house IVD notification form' and updated content to reflect regulation updates.
Added the 'Exempt Class 4 in-house IVD notification form' and updated content to reflect regulation updates.

