Recently published
This page was published on [date_placeholder].
Recently updated
This page was updated on [date_placeholder]. See page history for details.
Purpose
This guidance relates to the use of mandatory warning statements for labels of medicines that contain neuromuscular blocking agents, which present significant risk to patient safety.
Legislation
Neuromuscular blocking agents
Neuromuscular blocking agents are used to cause paralysis during anaesthesia.
Medicine administration errors of neuromuscular blocking agents present significant risk to patient safety due to potential for unintended paralysis, respiratory arrest, severe permanent harm (including physical and psychological harm) or even death.
Administration errors involving neuromuscular blocking agents in Australia can be caused by look-alike selection errors, i.e. where two medicines are similar in appearance resulting in the wrong medicine being chosen and administered.
Examples
To minimise look-alike medicine selection errors, we introduced mandatory warning statements for labels of medicines containing neuromuscular blocking agents.
An example of the warning statement is:

An example of a red warning statement label for an Australian medicine label for a product that contains neuromuscular blocking agents.
The warning statement has black text on a red background and reads, "WARNING: Paralysing agent".

An illustrated example of a blue medication box labelled "NMBA Injection" and the red warning label that reads "WARNING: Paralysing agent" in the top-left corner under, representing medicine packaging for a product that contains neuromuscular blocking agents.
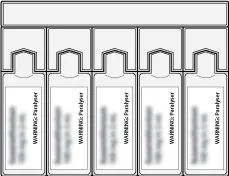
An illustration of 5 ampoules in a row containing a paralysing agent, with each ampoule depicted as a rectangular container with a rounded top and labelled with a shortened warning statement that reads, "WARNING: Paralyser".
Introduction of the warning statement
Introduction of the warning statement follows extensive consultation with anaesthetists, healthcare groups and medicine sponsors.
- Neuromuscular blocking agent containing medicines require a warning statement on the container and any outer packaging.
- Medicine sponsors were required to update their labels by September 2020, however you might still see medicines with old labels after this date, as this stock is gradually exhausted.
- Some sponsors have been granted a special consent to supply medicines with the old label for a time due to difficulty introducing the new labels. You can find out which medicines have been granted this consent in the Consent to supply database.
- Neuromuscular blocking agents are only available in hospitals for administration by health professionals. Patients are not expected to see these medicine labels.
Medicine ingredients that require the use of a warning statement
Medicines that contain the following ingredients must be labelled with the warning statement:
- suxamethonium
- pancuronium
- vecuronium
- atracurium
- rocuronium
- mivacurium
- cisatracurium.
Medicines that are already on the shelf or in dispensaries that do not include the warning statement may still be used.
Warning statement specifications and requirements
A warning statement must be on labels of the primary pack (outer carton) and the container (ampoule or vial).
For the primary pack and some containers, labels of medicines containing neuromuscular blocking agents have the warning statement WARNING: Paralysing agent in black text on fluorescent red or warm red background.
On very small containers, the warning statement on the container might be shortened to WARNING: Paralyser or Paralyser.
On plastic ampoules, the warning statement is likely to be shortened. It will also not be on a coloured background. This is due to the way these containers are manufactured.
The warning statement should alert health practitioners dispensing and administering the medicines of the potential harm that can be caused by these high-risk medicines and help distinguish these medicines from others.
The specified colours are consistent with the colours for neuromuscular blocking agents in the National Standard for User-Applied Labelling of Injectable Medicines, Fluids and Lines.
Old stock can still be supplied as long as it was released before 1 September 2020.
The requirements to include warnings on labels of medicines containing neuromuscular blocking agents reduce the chance of sponsors' medicines being incorrectly selected and administered by health professionals.
These requirements are included in Therapeutic Goods Order No. 91 - Standard for labels of prescription and related medicines (TGO 91), under section 6 and subsection 10(8A). Schedule 3 is the list of ingredients that must have these warning statements when used in medicines.
Medicine labels: Guidance on TGO 91 and TGO 92 contains information to assist you in meeting the requirements for neuromuscular blocking agents, including the specified background colour identifiers for 'fluorescent red' and 'warm red' (see sections 2.3.2 and 3.4.10).
The best practice principles, in Part 3 of the Guidance, should be followed. This means using the full warning statement wherever possible and placing it on the main label of outer packaging.
Page history
Title changed from 'Warnings on labels of medicines containing neuromuscular blocking agents' to 'Reducing safety risks with mandatory warning labels on medicines' as part of migration to new 'Guidance' content type:
- Consistent ‘Purpose’ heading.
- ‘Legislation’ section to clearly show which laws the Guidance relates to.
- ‘Page history’ section replaces document version history.
- New page navigation features.
- Updated page summaries.
- Complex images include long descriptions.
- New ‘Save as PDF’ feature.
Title changed from 'Warnings on labels of medicines containing neuromuscular blocking agents' to 'Reducing safety risks with mandatory warning labels on medicines' as part of migration to new 'Guidance' content type:
- Consistent ‘Purpose’ heading.
- ‘Legislation’ section to clearly show which laws the Guidance relates to.
- ‘Page history’ section replaces document version history.
- New page navigation features.
- Updated page summaries.
- Complex images include long descriptions.
- New ‘Save as PDF’ feature.

