Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
The Therapeutic Goods Administration (TGA) is warning consumers about the risks of importing and using unapproved peptide products that are promoted online.
The TGA continues to see peptide products imported through the Personal Importation Scheme. These products may pose significant risks to patient safety.
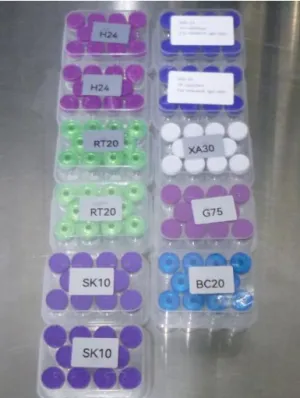
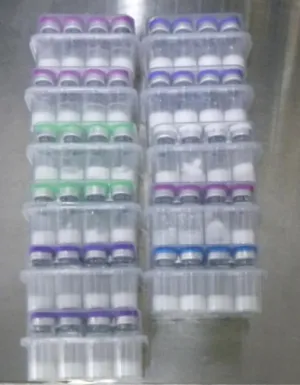
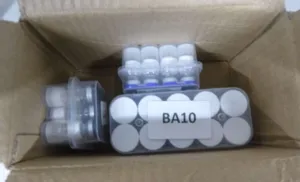
Products referred to the TGA by the Australian Border Force (ABF) are often poorly labelled. This makes it impossible to verify their contents, and mean they may be products that put your health at serious risk.
The TGA has identified imported products suspected to be peptides that:
- are not included in the Australian Register of Therapeutic Goods (ARTG)
- are supplied as powders or injectables in unmarked vials, and
- are not clearly labelled and therefore cannot be identified, are marked only with codes or abbreviations, or are missing essential information such as the name and concentration of the active ingredient or dosing instructions.
Importing unapproved peptide products under the Personal Importation Scheme is permitted only if the goods are clearly and accurately labelled to identify the therapeutic good. Examples of insufficiently labelled, unsafe or non-compliant products are provided below.
Professor Anthony Lawler, Deputy Secretary of the Department of Health, Disability and Ageing and head of the TGA, said " Australians should be very cautious about buying peptide products online, particularly from overseas websites or through online platforms or social media.
“Many of these products are unapproved, poorly labelled and not subject to Australian safety or quality standards. If you don’t know exactly what’s in the vial, where it was made or whether it’s sterile, you could be putting your health at serious risk. You could also incur financial loss if the product is intercepted at the border and cannot be lawfully released.”
Professor Robyn Langham AM, the TGA’s Chief Medical Advisor, said “the biggest risks with unapproved peptide products are not knowing what’s in the vial. There is no way of knowing how much of the peptide is in there, whether the contents are sterile or whether there are other contaminants or toxins in there that you risk administering to yourself.
“Unmarked vials, code‑only labels, or missing ingredient and dosing information are major red flags. To reduce your risk, you should buy therapeutic goods only from a legitimate Australian pharmacy or supplier, and talk to your doctor or another health professional, especially before using injectable products.”
Consistent with TGA requirements, these vials will not be released under the Personal Importation Scheme.
Actions we are taking
If peptide products do not meet importation requirements due to poor or unclear labelling, we will notify the ABF to seize and destroy these products when they are intercepted at the border.
Information for consumers
The TGA continues to provide education for consumers by:
- publishing safety advice about the risks of importing, compounding and supplying unapproved peptide products
- encouraging people using medicines bought online or from overseas to check TGA safety alerts and talk to a health professional, and
- advising consumers to be particularly cautious when buying medicines or medical devices online, including through online platforms or social media.
Reporting problems
If you experience an adverse event, please report it to the TGA. This helps us to monitor and respond to emerging safety concerns.
If you suspect non-compliance in relation to therapeutic goods, you can report a perceived breach or questionable practice to the TGA.
You can also report using:
| Phone: | 1800 020 653 |
| Email: | info@tga.gov.au |
Product images
|
|

|

|

|

|

| |
|

|

