Recently published
This page was published on [date_placeholder].
Recently updated
This page was updated on [date_placeholder]. See page history for details.
Purpose
As listed medicines are not evaluated, the Therapeutic Goods Administration (TGA) uses a variety of mechanisms to help ensure that they are of high quality and used safely by consumers. One of these mechanisms is that listed medicines must only use indications drawn exclusively from a list of pre-approved 'permitted indications' and must comply with any requirements for the use of these indications in listed medicines.
This guidance provides information for sponsors on:
- what permitted indications are, including terminology and structure
- the use of permitted indications for listed medicines; and
- how to apply for inclusion of new indications on the permitted indications list.
For further information on the regulation of complementary medicines in Australia, please refer to the Australian Regulatory Guidelines for Listed Medicines and Registered Complementary Medicines.
Legislation
Permitted indications for listed medicines
Listed medicines are considered 'low risk' and are not individually evaluated before they are entered in the Australian Register of Therapeutic Goods (ARTG). Upon submission of a listing application, applicants must certify that their medicine is eligible for listing and complies with all applicable regulatory requirements.
As listed medicines are not evaluated, the Therapeutic Goods Administration (TGA) uses a variety of mechanisms to help ensure that they are of high quality and used safely by consumers. One of these mechanisms is that listed medicines must only use indications drawn exclusively from a list of pre-approved 'permitted indications' and must comply with any requirements for the use of these indications in listed medicines.
This guidance provides information for sponsors on:
- what permitted indications are, including terminology and structure
- the use of permitted indications for listed medicines; and
- how to apply for inclusion of new indications on the permitted indications list.
What are permitted indications?
'Indications' are statements that describe the specific therapeutic use1 for a medicine. They describe a medicine's claimed purpose or health benefit, for example: 'relieves coughs'. Indications can be specific (e.g. refer to a named non-serious condition); or non-specific (e.g. refer to general health maintenance).
Indications available for use for listed medicines are included in a 'list of permitted indications' which is contained in the Therapeutic Goods (Permissible Indications) Determination (the Permissible Indications Determination). The list of permitted indications is maintained by the TGA and provides a comprehensive list of indications currently accepted for use in listed medicines, provided appropriate evidence is held by the medicine sponsor.
Consistent with their low risk status, listed medicines may only use low level indications that will not lead to their unsafe or inappropriate use. Permitted indications have been assessed against a set of criteria and determined to be appropriate for listed medicines. These criteria are intended to ensure that permitted indications will only cover (and listed medicines will be limited to making) indications relating to:
- health maintenance
- health enhancement
- prevention of a non-serious vitamin or mineral dietary deficiency; or
- certain non-serious2, self-limiting diseases, ailments, defects or injuries.
To be consistent with their low risk status, regulatory requirements are placed on the use of certain indications in listed medicines. These are also specified in the Permissible Indications Determination (see Requirements relating to the use of a permitted indication).
Does your product make therapeutic indications, cosmetic or food claims?
Some products are used in a similar way to therapeutic goods but are actually regulated as cosmetics or foods. The list of permitted indications includes a number of indications that could be used for therapeutic goods, cosmetic products or foods, depending on such things as the product's ingredients, presentation and route of administration3. Potential examples of such indications could be:
- 'Maintain/support hair strength' could be used for an oral therapeutic good, such as a vitamin/mineral supplement, while a similar indication might be used for a topical hair product presented as a cosmetic e.g. hair conditioner4.
- 'Enhance bone density' could be used for an oral therapeutic good, such as a mineral supplement, while a similar indication might be used for a food5.
- 'Decrease/reduce/relieve skin dryness' could be used for an oral therapeutic good, while a similar indication could be used for a topical moisturiser presented as a cosmetic.
Just because an indication is in the list of permitted indications does not mean that the use of that indication makes a product a therapeutic good. Further, inclusion of an indication in the list does not mean that only therapeutic goods can use that indication. Before listing a medicine in the ARTG, the sponsor should satisfy themselves that their product is a therapeutic good. Refer to the Australian Regulatory Guidelines on Complementary Medicines: Complementary medicines interface issues for information to help you determine whether or not your product is a therapeutic good.
Components of a permitted indication
Permitted indications have a consistent structure and terminology to describe the therapeutic uses for listed medicines.
All permitted indications contain a therapeutic action and a therapeutic target. In general, permitted indications form a complete sentence. However, in some circumstances an indication may contain an action and a target in a single word, for example: 'antiemetic' and 'laxative'.
Some indications also include an action or target qualifier to ensure that they are appropriate to be used for low risk listed medicines. Where an indication includes an action or target qualifier, it must be included on your medicine label (see Figure 1 and Table 1).
Figure 1 - Permitted indication structure
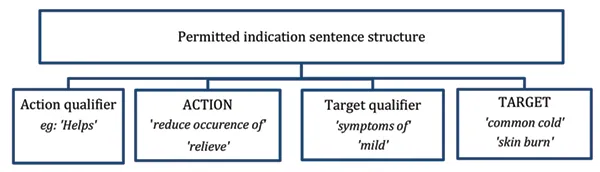
This image shows the structure of a permitted indication sentence. It consists of four components arranged from left to right:
- Action qualifier (e.g., 'Helps')
- ACTION ('reduce occurrence of' or 'relieve')
- Target qualifier ('symptoms of' or 'mild')
- TARGET ('common cold' or 'skin burn')
These components are presented in boxes connected by a single line, illustrating how they form a complete permitted indication sentence when combined.
| Indication component | Explanation | Use in permitted indications | Examples |
|---|---|---|---|
| Action | The therapeutic action or effect of a medicine | Always included | 'Reduce/relieve' 'Reduce occurrence of' 'Maintain/support' 'Enhance/improve' |
| Target | Either a: physiological / psychological factor or process; or | Always included | 'Bone strength' 'General health' 'Eye health' 'Headache' 'Excess mucous' |
| Action qualifier | Terms that ensure the action is suitable for a low level indication. They often specify the effectiveness of a medicine. | Sometimes included | 'Temporarily' 'Help' |
| Target qualifier | Terms that ensure the indication is not referring to a restricted representation (that is a serious form of a disease, disorder or condition). | Sometimes included | 'Mild' 'Symptoms of' 'Healthy' 'Medically diagnosed' |
Note that sponsors can modify a selected permitted indication using a set of pre-approved 'permitted indication qualifiers' to align with the supporting evidence they hold for their medicine. In the case of indications for which a sponsor holds evidence of traditional use, the sponsor must select a 'Traditional context qualifier' to identify the traditional paradigm in the ARTG entry and on the medicine label. Please see Selecting permitted indication qualifiers for your permitted indication for more information.
Product statements and claims that are not indications
Statements or claims that do not describe a therapeutic use are not included in the permitted indications list. Some examples are provided below.
- Marketing statements that are not related to the therapeutic use of the product
For example:
- 'Contains ingredients of plant origin'
- '20% more tablets, same price'
- Claims relating to the effectiveness of the product
For example:
- 'Improves X by 10%'
- 'Results in 2 weeks'
- 'Fast acting formula'
- 'Water resistant'
- Directions for use statements
For example:
'When taken three times a day'
- Structure, function, and/or mechanism of action statements that do not include an action, target or a therapeutic use
For example:
- 'X is a component of bone'
- 'X is used in the production of ATP'
While structure, function and mechanism of action statements are generally not therapeutic indications, in some instances they can be considered to imply a therapeutic use. A therapeutic use may be implied where the message conveyed to the consumer suggests a health benefit, that is, that the product is intended to influence, inhibit or modify a physiological process. Where this is the case, an indication of similar intent must be included in the ARTG entry.
Examples of structure, function and mechanism of action statements that imply a therapeutic use and possible permitted indication with similar intent are provided below.
For example:
- 'Calcium is a natural component of bone' (potential ARTG indication 'Maintain support health').
- 'X is a component of skin cell membranes' (potential ARTG indication 'Maintain/support healthy skin structure').
- 'X is a co-factor for mitochondrial function' (potential ARTG indication: 'Maintain/support energy production').
Product statements and claims that are not indications are not required to be included on the ARTG entry for the medicine in order to be used on product labels or other advertising. However, you must hold evidence (and be able to provide this evidence if requested) to support any statement/claim made for your medicine and comply with all relevant advertising requirements including the Therapeutic Goods Advertising Code (the Advertising Code).
Requirements for using permitted indications
To ensure that permitted indications are suitable for listed medicines, the Permissible Indications Determination includes 'requirements' for their use for listed medicines. As a sponsor, you must ensure that you comply with all 'requirements' that apply to the use of the permitted indications you select for your listed medicine. The Therapeutic Goods Act 1989 (the Act) allows for cancellation of a medicine from the ARTG if the requirements are not met.
Evidence requirements
At the time of listing a medicine in the ARTG, sponsors must certify that they hold evidence to support any indications and claims made about their medicine. Information on the types of evidence required to support scientific and traditional indications for listed medicines can be found in the Evidence guidelines or the Australian regulatory guidelines for sunscreens (ARGS).
The Permissible Indications Determination specifies the type of evidence that must be held by sponsors to support the use of each permitted indication for listed medicines. There are four categories of permitted indications based on the type of evidence required to support their use.
The types of indications and evidence sources are described in Table 2 below.
| Type of indication | Type of evidence |
|---|---|
| Scientific indications | Must be supported by scientific evidence, such as clinical studies or systematic reviews, for example: 'Help maintain/support bone mineralisation'. |
| Traditional indications | Must be supported by evidence of traditional use in a recognised paradigm outside modern conventional medicine. These include indications that can be used across different traditional paradigms, for example: 'Blood cleanser/purifier'. |
| Traditional Chinese medicine indications | Must be supported by evidence of traditional use within traditional Chinese medicine (TCM). These indications use specific terminology used in TCM, for example: 'Traditionally used in Chinese medicine to disseminate Lung Qi'. |
| Traditional Ayurvedic indication | Must be supported by evidence of traditional use within Ayurvedic medicine. These indications use specific terminology used in Ayurvedic medicine, for example: 'Traditionally used in Ayurvedic medicine to relieve aggravated Vata'. |
Note that some indications can be supported by scientific evidence or evidence of traditional use, for example: 'Improve/enhance digestion' and 'Traditionally used in Western herbal medicine to improve/enhance digestion'. For these indications the type of evidence is marked as a 'scientific or traditional'.
In preparing the Permitted Indications Determination, the TGA has not considered whether each indication is supported by evidence for each permitted ingredient or combination of ingredients.
As a sponsor of a listed medicine, it is your responsibility to ensure that your product meets all the requirements for listing, including that you hold evidence for all indications made for your medicine.
Other indication requirements
Other requirements included in the Permissible Indications Determination specify circumstances when individual indications can or cannot be used or specify conditions that must be met to use the indication for a listed medicine. For example, requirements may:
- specify a vulnerable population for which the indication is not suitable, such as children
- specify an advisory statement that must be included on product labels
- require that the product presentation does not imply or refer to a serious form of a disease (i.e. a restricted representation); and/or
- specify that a qualifying term(s) must be used on medicine labels, for example where an indication is qualified to only refer to a 'mild' form of a disease.
For examples of specific indication requirements see Table 3.
| Indication | Type of evidence | Other requirements |
|---|---|---|
| Antipyretic/Antihydrotic /febrifuge/relieve mild fever/reduce body temperature/body cooling | Scientific or Traditional | Label statement: If symptoms persist, seek the advice of a healthcare professional Label statement: Not to be used in children under 5 years |
| Helps prevent dietary (state vitamin/mineral/nutrient) deficiency | Scientific | Label statement: Vitamins and minerals can only be of assistance if dietary intake is inadequate OR Vitamin and/or mineral supplements should not replace a balanced diet |
| Helps enhance/promote preconception health | Scientific or Traditional | Product presentation must not imply or refer to infertility. Label statement: Advise your doctor of any medicine you take during pregnancy, especially in your first trimester |
| Balance Yin and Yang | Traditional Chinese medicine | Label statement: Please seek the advice of a traditional Chinese medicine practitioner to ensure this medicine is right for you. |
| Trophorestorative/restore organ health | Traditional | Product presentation must not imply or refer to disease in any body organ. |
| Decrease/reduce/relieve symptoms of mild arthritis/mild osteoarthritis | Scientific or Traditional | Label statement: If symptoms persist, seek the advice of a healthcare professional Product presentation must only refer to mild joint symptoms. Product presentation must not imply or refer to bone disease or disorders e.g. rheumatoid arthritis, juvenile arthritis, debilitating osteoarthritis, osteoporosis |
Requirements relating to using TCM and Ayurvedic permitted indications
TCM and Ayurvedic indications have been included in the list of permitted indications in recognition that traditional terminology is required for persons familiar with the these paradigms to use the medicines appropriately.
However, TCM and Ayurvedic terminology are not likely to be understood by average consumers. Therefore, it is a requirement (in addition to the requirement to include a traditional context qualifier) that medicines using indications with paradigm specific terminology include an advisory statement on their label with words to the effect of:
- 'Seek advice from a Chinese medicine practitioner if you are unsure if this medicine is right for you'.
- 'Seek the advice from an Ayurvedic medicine practitioner if you are unsure if this medicine is right for you'.
A sponsor may also choose to include an English version of the TCM or Ayurvedic indication (from the permitted indications list) in the ARTG and on the medicine's label alongside the traditional terminology, if the indication has the same meaning.
For example:
'Traditionally used in Chinese medicine to harmonise the stomach' (TCM) could, if appropriate, be accompanied by an English indication such as: 'Traditionally used in Chinese medicine to help maintain/support stomach health'.
Using permitted indications
Selecting permitted indications for your medicine
A searchable database of indications and their requirements is also available on the TGA Business Services website. Instructions on searching this database are provided below.
Searching for indications via the TGA Business Services website.
- Select 'Public TGA Information' from the left hand menu.
- From the dropdown menu select 'Indications for listed medicines'.
- Enter the indication key word/or phrase you are looking for in the 'search field' and ensure that in 'all fields' is selected.
- Click 'Go'.
When you apply to list your medicine in the ARTG using the Electronic Listing Facility (ELF), you can select indications for your medicine from a searchable catalogue of permitted indications.
You are able to search the list of permitted indications by:
- body system or body part (e.g. cardiovascular system, respiratory system)
- evidence requirement (e.g. scientific or traditional)
- key word/phrase (e.g. itchy or cough); and
- the indication's unique computer code (e.g. BDEYDR-G-RE).
For more information on how to include your medicine in the ARTG, please see the Listed medicines application and submission user guide.
Selecting indication qualifiers for your permitted indication
When you enter your medicine in the ELF, you may also choose to select indication qualifiers from drop down lists to make a permitted indication more specific and align with the evidence you hold for your medicine.
You:
- must include indication qualifiers on your medicine label (or other advertising) if you have entered them in the ARTG7
- must only use the indication qualifiers from the Code Tables accessible via the TGA Business Services website- external site.
- must not use an indication qualifier on your medicine label (or other advertising) unless you have included the qualifier in your ARTG entry
- must not use an indication qualifier to modify a selected permitted indication if it infers or implies that the medicine is for the treatment of a serious disease.
The types of indication qualifiers available for selection are described in Table 4.
| Indication qualifiers |
|---|
Traditional context qualifier This specifies the traditional paradigm and is a mandatory qualifier for indications where the sponsor holds evidence of traditional use in any paradigm. For example: 'Traditionally used in Western herbal medicine...'. The traditional context qualifier is not applicable for indications supported by scientific evidence. |
TCM pattern qualifiers This is an optional qualifier for TCMs and specifies the underlying pattern that causes rise to the symptoms included in the selected indication. For example: '...in spleen Qi deficiency pattern'. |
Population qualifiers This is an optional qualifier specifying the target population for the medicine. For example: '...in healthy individuals', '...in women', '...in men'. |
Time of use qualifiers This is an optional qualifier and indicates the time of therapeutic benefit for the medicine, for example: 'Increase energy levels throughout the day'; or the time of occurrence of a symptom e.g. 'Relieves muscle stiffness after exercise'. |
Figure 2 shows the use of a permitted indication with indication qualifiers.
Figure 2 - Use of permitted indications with indication qualifiers
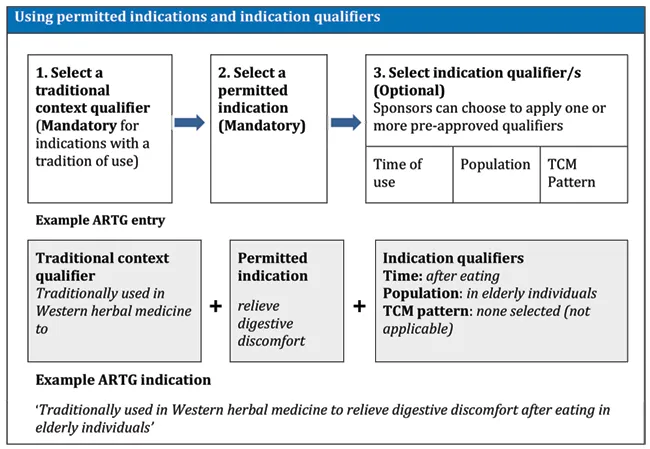
This image illustrates the process of using permitted indications and indication qualifiers. It consists of three main steps:
- Select a traditional context qualifier (Mandatory for indications with a tradition of use)
- Select a permitted indication (Mandatory)
- Select indication qualifier/s (Optional)
For step 3, sponsors can choose to apply one or more pre-approved qualifiers: Time of use, Population, and TCM Pattern.
The image also includes an example ARTG entry:
- Traditional context qualifier: "Traditionally used in Western herbal medicine to"
- Permitted indication: "relieve digestive discomfort"
- Indication qualifiers
- Time: after eating
- Population: in elderly individuals
- TCM pattern: none selected (not applicable)
The resulting example ARTG indication reads: "Traditionally used in Western herbal medicine to relieve digestive discomfort after eating in elderly individuals"
For each permitted indication, you can only select one of each of the four indication qualifier types described in Table 4. If you wish to select multiple qualifiers from within one of the indication qualifier groups (for example: select multiple population qualifiers) for your permitted indication, you will need to enter the indication multiple times in the ARTG.
For example:
A sponsor holds scientific evidence that their medicine is used to relieve dry skin in children and adults. The ARTG indications entered for this medicine could be:
- ARTG indication1: 'Relieve dry skin in children'
- ARTG indication2: 'Relieve dry skin in adults'
The indication included on the medicine label could be: 'Relieve dry skin in children and adults'
As a medicine sponsor, you must ensure that the safety of your medicine has been established for the target population (e.g. infant target population) that you choose to add to your permitted indication. Under provisions in the Act, a medicine that not safe for its intended use can be cancelled from the ARTG.
Selecting a traditional context qualifier
Indications that are supported by evidence of traditional use must identify the traditional paradigm in the ARTG entry as well as when used on labels and other advertising material.
If your supporting evidence is based on traditional use:
- you must include the relevant traditional context qualifier in the medicine's ARTG entry
- you must include the traditional context qualifier when used on your label and other advertising material.
When selecting a traditional context qualifier for a permitted indication, you must ensure that the indication is consistent and appropriate for the traditional paradigm. For more information refer to Permitted indications must be consistent with the relevant treatment paradigm.
| Evidence held by sponsor | Mandatory traditional context qualifier | Indication |
|---|---|---|
| Traditional use in Chinese medicine | Traditionally used in Chinese medicine | clear/expel stagnant Heat |
| Traditional use in Ayurvedic medicine | Traditionally used in Ayurvedic medicine | relieve aggravated Vata |
| Traditional use in Western herbal medicine | Traditionally used in Western herbal medicine | nourish the heart |
| Scientific evidence | Not applicable | Maintain/support bone mineralisation |
If your medicine includes indications supported by evidence of traditional use across different traditional paradigms, you must include the traditional context qualifier for each paradigm in the ARTG, even if the indications refer to the same health benefit. If the indications refer to the same health benefit the indication/s can be combined on your medicine label to form a single indication.
For example:
A sponsor holds evidence of traditional use to relieve nasal congestion in the traditional Chinese and Ayurvedic medicine paradigms. The ARTG indications entered for this medicine could be:
- ARTG indication1: 'Traditionally used in Chinese medicine to relieve nasal congestion'
- ARTG indication2: 'Traditionally used in Ayurvedic medicine to relieve nasal congestion'
The indication included on the medicine label could be: 'Traditionally used in Chinese medicine and Ayurvedic medicine to relieve nasal congestion'.
If the different traditional paradigms are used for different health benefits, you will need to include the indications separately in the ARTG entry and on your medicine label.
For example:
A sponsor holds evidence of Australian indigenous use to relieve nasal congestion and evidence of traditional Western herbal medicine use to relieve symptoms of common colds and flu. The ARTG indications entered for this medicine could be:
- ARTG indication1: 'Traditionally used in Australian indigenous medicine to relieve nasal congestion'
- ARTG indication2: ''Traditionally used in Western herbal medicine to relieve the symptoms of common colds and flu'
The indications included on the medicine label could be:
'Traditionally used in Australian indigenous medicine to relieve nasal congestion'.
'Traditionally used in Western herbal medicine to relieve the symptoms of common colds'.
See Use of permitted indications on medicine labels for more information on how to ensure your medicine label is compliant with the regulatory requirements.
Selecting TCM pattern qualifiers for your TCM permitted indication
TCM patterns are an optional indication qualifier and can be used by TCM sponsors to make TCM indications more specific and align with the evidence you hold for your medicine. TCM patterns have been included in recognition that traditional terminology is required for persons familiar with these paradigms to use the medicines appropriately.
If you select the traditional context qualifier 'Traditionally used in Chinese medicine' when entering your indication in ELF, you will be able to select traditional permitted indications and TCM specific permitted indications. In addition, you will be able to select a 'TCM pattern' qualifier to add to your permitted indication.
For example:
Traditionally used in Chinese medicine to 'dispel Wind Heat' (TCM indication) in 'externally contracted Wind-Heat pattern' (TCM pattern qualifier)
Sponsors using TCM permitted indications and TCM pattern qualifiers must ensure that the resulting combined indications are consistent with the Chinese medicine paradigm.
Using permitted indications on medicine labels and advertising
To allow sponsors some flexibility in label design you are not required to include permitted indications included in your medicine's ARTG entry 'word for word' on your medicine label and other advertising material unless there is a specific requirement for you to do so. However, the indication entered in the ARTG and medicine label (and other advertising material) must have the same meaning. This means that the indication in the ARTG and on the medicine label and other advertising material must have an equivalent therapeutic action and target.
As a sponsor of a listed medicine, you are responsible for ensuring that the overall presentation of your medicine (including the label and any advertising used by you or provided to any other person for their use) is compliant with the labelling order, the advertising requirements in the Act and the Advertising Code. Particular care is required when modifying the wording of indications to ensure that they remain compliant with the advertising requirements.
A medicine may be cancelled from the ARTG if the sponsor does not comply with the advertising and labelling requirements.
Some product names can imply a therapeutic indication. Where this is the case, the implied therapeutic indication must be included in the ARTG entry as a permitted indication as well as on the product label.
Acceptable variation of permitted indications
You can:
- change the structure or order of the components of a permitted indication
- use the therapeutic action terms included in a permitted indication interchangeably (e.g. decrease/reduce).
You must not:
- modify a selected permitted indication to give it a different, broader or more specific therapeutic target
- modify a mandatory qualifying term (e.g. 'mild', 'medically diagnosed') on your medicine label or other advertising material
- modify a selected permitted indication to infer or imply that a medicine is for the treatment of a serious disease
- modify a selected permitted indication to infer or imply that a medicine is for the prevention or cure of any disease.
For example:
*Note: The following are provided as examples only and are not an endorsement of the use of these indications. The overall presentation of your medicine and the evidence you hold must be considered in determining if indications have the same meaning.
Example 1: ARTG indication: 'Maintain/support gastrointestinal system health'
Label indication – may have same meaning: 'Maintains healthy gut'
Label indication – may have same meaning: 'Gastrointestinal health support'
Label indication – different meaning: 'Maintain healthy intestinal flora'
In this example 'intestinal flora' is a more specific target than the general indication of 'Maintains healthy gut' and therefore the meaning of the indication has changed.
Example 2: ARTG indication: 'Reduce/decrease mild muscle inflammation'
Label indication – may have same meaning: 'Relieves mild inflammation in muscles'
Label indication - different meaning: 'Relieves muscle pain'
In this example 'inflammation' and 'pain' are different targets and therefore the meaning of the indication has changed.
Example 3: ARTG indication: 'Decongestant/relieve nasal congestion
Label indication – may have same meaning: 'Unblocks the nose'
Label indication – may have same meaning: 'Clears the nasal passage'
Label indication – different meaning: 'Unblocks sinuses'
In this example 'sinus' is a different target to 'nose' and 'nasal passage' and therefore the meaning of the indication has changed.
Example 4: ARTG indication: 'Decrease/reduce/relieve symptoms of mild arthritis/mild osteoarthritis'
Label indication – may have same meaning: 'Relieves mild arthritis symptoms'
Label indication – different meaning: 'For the treatment of arthritis symptoms'
In this example, the ARTG indication includes the target qualifier 'mild', which must be included on the medicine label to have the same meaning. In addition, the indication refers to treatment, which is not appropriate for listed medicines.
'May' as a qualifier
In general, the action qualifier 'may' is not appropriate as an action qualifier for permitted indications as it can imply that the sponsor does not hold evidence of sufficient quality to support the use of the indication for their medicine. This is not consistent with the legislative requirement for sponsors to hold evidence for the indications they make for their medicine.
Combining permitted indications on medicine labels
You can enter multiple indications in your ARTG entry and combine these into complete sentences on your medicine label and other advertising (see Table 7 below).
When combining indications you must:
- comply with all requirements that apply to the individual indications
- use any mandatory action and target qualifiers such as mild, helps, temporary etc. in relation to the combined indication on the product label
- ensure that you hold evidence to support the combined indications.
You must not:
- combine selected permitted indications to give them a different, broader or more specific meaning
- combine selected permitted indications infer or imply that a medicine is for the treatment of a serious disease
- combine selected permitted indications to infer or imply that a medicine is for the prevention or cure of any disease.
| Indications included in the ARTG | Indications included on the Medicine label |
|---|---|
|
|
|
|
It is possible to enter multiple indications in a medicine ARTG entry that have different evidence bases, such as evidence of traditional use and scientific evidence. These indications must be entered individually in the ARTG so that the evidence base is clear. When using these indications on labels and other advertising material, particular care is required to ensure that they remain compliant with the advertising requirements and that the combination of indications is not misleading.
Linking indications referring to symptoms with diseases or conditions
Permitted indications contain a single 'target' and do not link general symptoms (e.g. 'relieves runny nose') to related conditions (e.g. 'relieves runny nose associated with common cold'). Instead, you can enter a number of indications referring to general symptoms and an indication referring to a disease or condition in your ARTG entry and combine these on your medicine label. An example is provided in Figure 3 below.8
When linking general symptom indications with an indication referring to a disease or condition, you must hold evidence for the selected symptoms for that disease or condition.
| Linked indication | ||
|---|---|---|
| Indication in ARTG | Decrease/ reduce/relieve symptoms of common cold | |
| Linked indications in ARTG |
| These symptoms can be linked to 'symptoms of common cold' on the medicine label |
These indications can be combined on the product label to form a simple sentence, as shown in the mock-up label in figure 3.
| Medicine information | |
Active Ingredient What this medicine is used for: | Warnings Other information Supplied by |
Applying for new indications
If you want to use an indication for your medicine that is not currently approved for use in listed medicines, you can apply for new indications to be added to the list of permitted indications. Applications must be submitted using the 'Indication and Qualifier application' form via the TGA Business Services website. An application fee applies.
The TGA will not evaluate evidence to support the use of a new proposed indication for a listed medicine at the time of application and in general there will be no requirement to submit supporting information. However, the TGA will assess whether the indication meets the Criteria for permitted indications described below.
The Permitted indication assessment tool has been developed to help you determine if a proposed indication is suitable to be included in the permitted indication list. This tool also shows what we will consider when making a decision to accept or refuse an application for a new indication. We recommend that you use the tool to self-assess whether your indication is suitable for inclusion in the permitted indications list before submitting it to us.
A decision by the Secretary (or a delegate) in relation to an application to add a new indication to the list of permitted indications is a reviewable decision. More information about requesting reconsideration of a reviewable decision can be found at Guidance for requesting reconsideration of an initial decision.
Criteria for permitted indications in listed medicines
The TGA will assess proposed indications against the following criteria to confirm that they are appropriate for use in low risk listed medicines. The indication must be:
- a 'low level' indication
- consistent with a treatment paradigm (for example, based on scientific or evidence of traditional use)
- capable of complying with the advertising requirements, including the Act and the Advertising Code when included on product labels and within promotional materials
- not included in a non-permitted indication list (if such a list is made).
Permitted indications must be 'low level'
Low level indications are those indications that are appropriate for medicines that are not assessed pre-market and will not lead to unsafe or inappropriate use of a medicine.
The eligibility requirements for permitted indications are based on a risk based approach to regulation. The risk factors relevant to therapeutic indications include, for example:
- whether the indications refer to a healthy person, or persons suffering from a disease, condition, ailment, defect or injury
- whether the indication relates to use in a vulnerable target population such as pregnant women, neonates or infants or the elderly
- whether the indication relates to a disease or condition that is potentially serious and as a result is inappropriate for self-diagnosis or self-management and/or the use of the medicine may delay consumers seeking medical treatment.
Products making low level indications pose a lower risk to consumers and it is appropriate that medicines with such indications are not individually evaluated by TGA before the medicine is included in the ARTG. Consistent with the risk-based framework, low level indications include indications for general health and wellbeing and self-diagnosable; self-manageable conditions; where a delay in medical treatment is unlikely to be detrimental to the consumer.
In certain circumstances an 'action qualifier' and/or a 'target qualifier' or specific indication requirement may be included in a proposed new permitted indication to ensure that the indication is low level.
What permitted indications can refer to
A proposed new indication may only refer to the following:
- Health maintenance
The indication can refer to the physiological effects of substances on normal growth, development or other functions of the body.
For example:
- 'Maintain/support intestinal good/beneficial/friendly flora
- 'Maintain/support eye health'
- 'Maintain/support heart health'
- Health enhancement
The indication can refer to beneficial effects of substances on the physiological and/or psychological state of the body above and beyond normal growth, development and functions of the body.
For example:
- 'Enhance/promote healthy digestion'
- 'Helps enhance/promote healthy liver function'
- Prevention of a dietary deficiency
The indication can refer to the prevention of vitamin or mineral dietary deficiency (not the prevention of diseases resulting from a physiological deficiency e.g. anaemia).
For example:
- Helps prevent dietary (state vitamin/mineral/nutrient) deficiency'
- A non-serious form of a disease, ailment, defect or injury
The indication can refer to a disease, condition, ailment, defect or injury that is self-diagnosable and/or self-manageable, where a delay in medical treatment will not be detrimental to the consumer. These indications may relate to:- reduction in risk, frequency, duration or severity of a non-serious form of a disease, condition, ailment, defect or injury
- relief of symptoms, without implying resolution or cure of the non-serious form of disease, condition, ailment, defect or injury.
For example:
- 'Relieve digestive discomfort'
- 'Relieve symptoms of mild upper respiratory tract infections'
- 'Reduce duration of symptoms of mild tonsillitis'
What permitted indications cannot refer to
A proposed new indication must not refer to any of the following:
- Prevention, cure or alleviation of any form of a disease, ailment, defect or injury
The indication must not refer to or imply the prevention, cure or alleviation of any disease, ailment, defect or injury. In this context, these terms are considered to imply the complete resolution of the disease, ailment, defect or injury.
For example:
- 'Cures eczema'
- 'treats dehydration'
- 'Prevents cold sores'
In certain circumstances, it may be possible to include an action qualifier to make an indication less definitive and meet the low-risk criteria, for example; 'Helps reduce occurrence of muscle cramp'. The qualifying term 'helps reduce the occurrence of' is less definitive than 'prevention' and is considered appropriate for use in the permitted indications list for certain indications.
Qualifying terms can only be used where the disease, ailment, defect or injury is considered non-serious and the consumer can self-diagnose, self-manage and self-assess the effectiveness of the medicine.
- A prohibited representation
The indication must not refer to a prohibited representation. Prohibited representations are defined in the Advertising Code.
As an exception to this criterion, the list of permitted indications includes indications covered by the prohibited representation exemption for broad spectrum sunscreens with SPF of 30 or higher (in relation to prevention of skin cancer).
- A restricted representation
The indication must not include any reference or implication to a restricted representation. A restricted representation is a 'serious form' of a disease, condition, ailment or defect specified in the Advertising Code.
As an exception to this criterion, the list of permitted indications includes the indications covered by the substance-based restricted representation exemptions linked to vitamin D and calcium (referring to osteoporosis) and folic acid (referring to neural tube defects) on the basis of their public health importance, safe history of use and well-established evidence base.
All other indications that refer to a restricted representation will require assessment and approval through a pre-market assessment pathway.
Use of 'action qualifiers' and 'target qualifiers' to ensure that proposed indications are low level
In some cases, the TGA will accept a proposed new permitted indication that makes a reference to what could considered a serious form of a disease, condition, ailment or defect if it is appropriately modified with an 'action qualifier' and/or a 'target qualifier' so that it meets the criteria for a low level indication.
These 'qualifying terms' can only be used to modify an indication where it is possible for a consumer to self-diagnose and self-manage the symptoms of the condition.
Note that despite the general principles outlined below, conditions that require ongoing supervision by a medical practitioner are not suitable for listed medicines.
Examples of 'action qualifiers' and 'target qualifiers' are provided in Table 8 and Table 9 below.
| Action qualifiers | |
|---|---|
| 'Helps' | The action qualifier 'helps' must be included in a permitted indication where:
|
| 'Temporary' | The action qualifier 'temporary' may be included in a permitted indication to ensure that an indication remains low risk and will not delay a consumer seeking medical attention if their symptoms persist. |
| Target qualifiers | |
|---|---|
| References to 'mild' forms of conditions | The target qualifier 'mild' may be used to qualify an indication to make it clear that the indication is only referring to a non-serious form of a disease, condition, ailment or defect, for example: 'mild arthritis', 'mild eczema'. Note that the use of the target qualifier 'mild' cannot be used for conditions that require ongoing medical management, for example: benign prostatic hyperplasia, rheumatoid arthritis. |
| References to 'medically diagnosed' conditions | For a limited number of conditions, it may be appropriate for a person to self-assess and/or self-manage the symptoms of their condition after an initial diagnosis has been made by a medical practitioner (including where other more serious diseases and conditions have been ruled out), for example: irritable bowel syndrome, shingles, cystitis. In these circumstances, the indication target (that is, the disease, condition, ailment or defect) may be qualified with 'medically diagnosed'. A required label statement will be 'If symptoms persist or worsen, consult your doctor'. Serious diseases, conditions, ailments or defects that require ongoing supervision by a medical practitioner cannot be qualified by 'medically diagnosed' as they are not suitable for listed medicines. E.g.: asthma, benign prostate hyperplasia. |
Permitted indications must be consistent with the relevant treatment paradigm
The indication must be consistent with a recognised treatment paradigm and the evidence base (scientific or traditional use) required to support its use.
Your scientific indication must not:
- use traditional medicine terminology, such as: 'alterative' or 'cholagogue'.
Your traditional indication must not:
- refer to diagnostic findings or anatomical, physiological or pharmacological effects that are not envisaged within the specified paradigm, for example: 'raise haemoglobin levels'
- use specialist terminology that belongs to a different paradigm or mainstream medical terminology, for example: 'damp heat' is a specific Chinese medicine term and would be inappropriate for an Ayurvedic medicine. Reference to blood glucose would be inappropriate for either paradigm
- include indications or use terminology that requires scientific substantiation, for example: 'assists to increase bone density by 10%'
- refer to conditions that cannot be diagnosed within the specified paradigm, for example: 'Traditionally used in Chinese medicine to increase bone mineral density' is inappropriate as increased bone mineral density cannot be monitored or determined without conventional medical intervention.
Permitted indications must be capable of complying with the advertising requirements and the
Therapeutic Goods Advertising Code
The indication must be capable of complying with the advertising requirements set out in Part 5-1 of the Act and the Advertising Code when included on the product label or in promotional materials.
For example, an indication when linked to ingredients or product formulation must not:
- mislead, or be likely to mislead consumers
- contain any implication that the medicine is infallible, unfailing, magical, miraculous, or that it is a certain, or guaranteed cure
- contain any claim, statement or implication that it is effective in all cases of the condition.
To not be considered misleading, the indication must be able to be supported by currently available scientific knowledge or a documented tradition of use. The indication cannot be impossible to be proven true, for example: 'Improves the IQ of your unborn child'.
An indication must not:
- imply definitive efficacy
- infer superiority over an evaluated product, for example: an assessed listed medicine, a registered complementary medicine, OTC or prescription medicine.
Other considerations
Public health and safety policies or principles
Public health messages are an investment by the Government in the promotion of public health and safety. Because listed medicines are not assessed by the TGA pre-market, indications that conflict with current public health policies will not be accepted for inclusion in the permitted indications list. The Government's priorities in public health messaging change, depending on trends and needs within the community. As such, it is not possible to provide an exhaustive list of indications that are unacceptable due to a conflict with public health messaging. However, some examples include:
- smoking
- vaccination/immunisation
- substance abuse, such as alcohol or narcotics
Inappropriate use of medicines
Consideration will also be given to whether an indication encourages inappropriate behaviour or inappropriate use of medicines. For example: emetics, which pose a significant hazard if used incorrectly and also pose significant potential for misuse e.g. bulimia.
Note that where there is a public health need or to ensure that listed medicines are used safely, a mandatory requirement will apply to the use of certain indications that products and promotional material cannot be directed to certain vulnerable population subgroups. See Requirements relating to the use of a permitted indication for more information.
Applying for new indication qualifiers
You can apply through the ELF to TGA for additional indication qualifiers to be available for selection. Applications must be submitted using the 'Indication and Qualifier application' form. This is available from the 'Applications' drop-down menu after you logged into your sponsor portal via the TGA Business Services website. Proposals for new indication qualifiers can only be for:
- a traditional context (with a document history of use), for example: 'Traditionally used in Western herbal medicine'
- a target populations, for example: 'in elderly males', 'in healthy adults'
- a TCM pattern, for example: 'in/of interior excess pattern', 'in/of Liver Fire pattern'
- time of use, for example: 'after exercise', 'after eating'. Time of use qualifiers are not intended to be directions for use statements, such as 'when taken twice a day' and are also not intended to imply an additional disease, disorder or condition to what is covered in the selected indication, such as 'when suffering with a cold'.
In principle, indication qualifiers should make an indication more specific and should not broaden the application of the indication.
Indication qualifiers cannot:
- have the potential to increase the risk of the indication, for example: include a vulnerable target population
- include or imply an additional therapeutic use by including a condition or symptom, for example: 'in times of anxiety' or 'associated with the common cold'
- relate to a direction for use, for example: 'when taken daily'
- refer to claims of efficacy, for example: '25% more effective
- be marketing claims, for example : '25% more potent formula'.
Appendix 1: Permitted indication assessment tool
| Answer the following questions in relation to the proposed indication | Yes | No |
|---|---|---|
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
If you answered NO to any question numbered 1 to 8 the indication is NOT SUITABLE. If you answered YES to questions 1 to 8, continue with questions 9 to 17. | ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| ||
| If you ticked YES to any question numbered 9 to 17, the indication is NOT SUITABLE. |
Footnotes
- 'Therapeutic use' is defined in section 3 of the Therapeutic Goods Act 1989.
- Note that the term 'serious' as used in this document has the same meaning as in the Therapeutic Goods Advertising Code 2015, which defines that term as meaning forms of those diseases, conditions, ailments or defects which are generally accepted either to not be appropriate to be diagnosed or treated without consulting a suitably qualified healthcare professional or to be beyond the average consumer's ability to accurately evaluate and treat safely without regular supervision by a qualified healthcare professional (e.g. cardiovascular diseases, haematological diseases).
- E.g, Order that Goods are Therapeutic Goods No. 2 of 1999 declares that products promoted for cosmetic purposes when promoted for oral consumption are, for the purposes of the Act, therapeutic goods.
- Please refer to the National Industrial Chemicals Notification and Assessment Scheme- external site for information on the regulation of cosmetics.
- Please refer to Food Standards Australia New Zealand- external site for information on health claims for foods.
- Listed complementary medicines may only contain low risk ingredients permitted for use in listed medicines as included in the Permissible Ingredients Determination. See Overview of listed complementary medicines for more information.
- Note that you do not need to include all indications entered in your medicine’s ARTG entry on your
medicine label. - Note that indications referring to general symptoms can be included in the ARTG as a standalone indication and are not required to be linked to a named disease or condition.
Page history
Original publication.
Original publication.

