Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
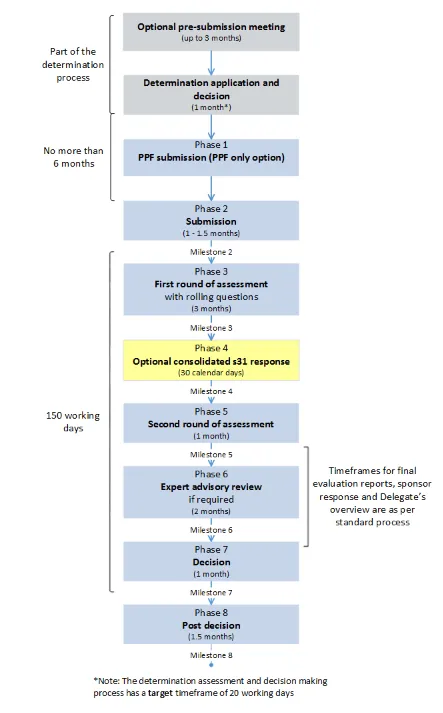
This guidance assists sponsors in understanding the process for submitting applications for priority registration. It outlines the key differences between the priority registration process and the standard prescription medicines registration process. Therefore this guidance should be read in conjunction with guidance on the standard registration process for prescription medicines.
This guidance should be read in conjunction with guidance on the prescription medicines registration process.