Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.

Foreword
The Therapeutic Goods Administration (TGA), as part of the Australian Department of Health, supports the Department's strategic priorities for an affordable, accessible, efficient and high-quality health system. It achieves this through effective regulation that maintains and improves the health and safety of the Australian community, while reducing compliance burdens.
Internationally, the TGA is a highly regarded regulator of medicines, medical devices, blood, cell and tissue products. We work closely with other regulators, both in comparable and developing health systems, as well as the World Health Organization (WHO). Our international engagement contributes to improved regional and global public health.
In an increasingly connected world, Australians want timely access to a range of established and emerging products available to treat health conditions. Both Europe and the United States of America (USA), with their large populations, generally attract manufacturers to these markets first. Consequently, many therapeutic products are authorised for use first in either the European Union (EU) or in the USA. New partnerships that have arisen from the United Kingdom's (UK) departure from the EU, the significant EU reform agenda and other emerging global changes are important considerations to ensure that our regulatory framework continues to support the interests of the Australian public to access the products manufactured in these markets. Australia will continue to make sovereign decisions, using our scientific expertise and skills to ensure that therapeutic products authorised for use in Australia are relevant to our context and health system, but we will increasingly collaborate with other regulators to expedite and inform these decisions.
Regulation of therapeutic goods occurs across the product life cycle and includes the approval, supply, manufacture, product safety surveillance and accurate advertising of therapeutic goods.
The TGA's International Engagement Strategy 2021-2025 describes how working with our international regulatory counterparts will benefit Australians through a more globally aligned regulatory framework. Reduced regulatory burden on industry, a fit for purpose regulatory system that is responsive to the latest regulatory science developments and enhanced global identification of safety signals leads to improved access to the latest health products and better safeguards for the Australian community.
We will achieve this by focusing on four Strategic priorities and a framework outlined under our Goals and activities.
Adj. Professor John Skerritt
The changing nature of TGA's role
The TGA's role as a regulator involves market assessment of a sponsor's product application through to proactive monitoring across the product lifecycle. This includes working with industry and other regulators at all stages to streamline regulatory processes. There is an increasing role to provide greater education and support, including to the public, which has been highlighted during the COVID-19 response.
Our alignment with European regulations for medical devices, whilst taking into account the significant reforms being implemented the by the European Commission, is also informed by identifying the most appropriate regulatory settings for Australia. In some circumstances this instead means aligning and adopting International Medical Devices Regulatory Forum (IMDRF) guidance or harmonising with US Food and Drug Administration (FDA) guidance. We will continue to contribute to the IMDRF Strategy across 2021-2025 to identify opportunities for greater alignment of pre- and post-market requirements and regulatory reviews, particularly for emerging technologies. We will also be taking a greater role in working with other countries on supply chain resilience.
Medicines regulation is becoming increasingly complex, requiring a constant evolution in regulatory sciences. TGA's leadership role in forums such as the International Coalition of Medicines Regulatory Authorities (ICMRA) allows us to better safeguard public health through greater cooperation on new international initiatives and common regulatory challenges. This forum facilitates information exchange and collaboration on issues of mutual interest, enables cooperation and promotes convergence of regulatory approaches for pharmaceutical approvals and monitoring.
Emerging regulatory challenges and opportunities
Regulators will be required to continue to work together globally to share ideas and discuss solutions in response to a range of emerging regulatory challenges and opportunities. Emerging regulatory challenges for medical devices often arise from technologies that incorporate new features and functions to enhance their performance. Using artificial intelligence (AI), software and digital technologies means that some devices are now capable of continuously adapting to improve their clinical performance specifications not fixed as with hardware medical devices. Our regulations, whilst flexible to support emerging trends, need to safeguard patient harms in relation to accessibility, cybersecurity, interoperability, data integrity and security. Ensuring the safety, quality and performance of the increasing demand for personalised implants requires a balance between necessary regulatory oversight, without impeding innovation.
The increasing complexity and technological sophistication of biological products, including cell and gene-based therapies, vaccines and regenerative medicine therapies also require innovative approaches to regulate. Novel technologies for advanced manufacturing of these products will also require advances in the TGA's scientific capabilities to inform regulatory oversight.
Implementation of global track and trace systems for medicines and medical devices will help protect public health by improving information sharing in case of quality defects, reducing shortages, contributing to the fight against falsified medicines and medical devices and supporting our vigilance activities. The TGA is working with stakeholders on improved ways to track and trace medicines and medical devices including opportunities for interoperability with the broader healthcare sector.
TGA's response to COVID-19
International regulatory collaboration has increased significantly to respond to the COVID-19 pandemic. The TGA streamlined the authorisation process for certain therapeutics, vaccines and medical technologies to support these efforts. Post-market vigilance and monitoring activities have also been enhanced to manage emerging safety concerns and ensure public confidence in the response to the pandemic, including the immunisation program.
The TGA has also engaged with other regulators and suppliers to accelerate approval for new medical devices including COVID-19 test kits, ventilators and personal protective equipment. The TGA continues to contribute to the global pandemic as Vice-Chair of ICMRA, and leading work on vaccines confidence through the COVID-19 vaccines pharmacovigilance network through its work with the WHO on clinical trial requirements for vaccines.
The TGA's role in supporting public health emergencies is critical. The surge of new products developed in response to the COVID-19 pandemic created unique challenges. Expectations for quality, safe and effective products have emphasised the critical role regulation of therapeutic goods plays in safeguarding and maintaining public health.
Based on the lessons learnt during the pandemic, the TGA and other regulators will continue to consider ways to leverage current processes and networks for future health emergencies, including:
- Rapid sharing of information on emerging products, risks, and supply chain concerns amongst regulators
- Increased regulatory agility to allow new products to market whilst maintaining safety, quality and efficacy requirements
- Establishing new protocols to enable remote inspections to ensure good manufacturing, through a mix of digital media and reliance on certified counterparts
- Enhancing communication channels to allow for more timely sharing of adverse events and compliance matters
Strategic priorities
Over the next four years, we will focus on four strategic priorities to foster international partnerships to support and enhance our
- Global policy alignment
- Pre-market global collaboration
- Post-market global monitoring
- Regional regulatory capabilities
Goals and activities
The following goals support the four strategic priorities (see Figure 1):
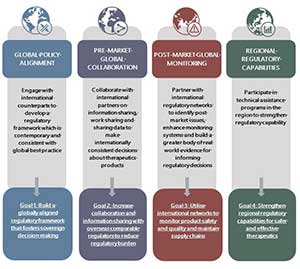
Figure 1. Alignment of goals with our four strategic priorities and objectives.
Goal 1 Build a globally aligned regulatory framework that fosters sovereign decision making
We will engage with international counterparts to develop a regulatory framework which is contemporary and consistent with global best practice.
A globally aligned regulatory framework reduces the regulatory burden on industry whilst balancing the need to ensure products brought to market are of high quality, safe, and effective. It also supports alignment and an expedited response to health emergencies. A globally aligned regulatory framework allows regulators across the world to come together to determine how to get new, safe, effective and quality therapeutics, vaccines and medical devices to patients as quickly as possible.
In response to the COVID-19 pandemic, there is unprecedented cooperation, collaboration, information sharing and understanding among international regulators. The global regulatory system was pressure tested and many lessons learnt. An opportunity exists to leverage this information and build on previous work to converge, align regulatory requirements between countries and develop a globally aligned regulatory framework that spans the lifecycle of therapeutic products.
Engaging with our international counterparts helps the TGA maintain an effective regulatory framework that is contemporary and consistent with global best practice. It allows us to keep pace with emerging technologies and public expectations for access to new and innovative therapeutic goods. Enhanced international engagement is critical if we are to influence the direction of current trends while maintaining independent decision making.
Summary of Activities
- Participate in or lead international projects on the alignment of policy, standards and guidelines for therapeutic products in response to emerging trends
- Continued involvement in developing internationally aligned regulatory policy responses to public health emergencies
- Manage regulatory developments for medical devices and innovative technologies by working with international forums to develop timely and appropriate guidance
- Participate in International initiatives which align identification of medicines and medical devices throughout the supply chain, thereby reducing shortages and improving safety
- Further develop mutual reliance frameworks to reduce the regulatory burden on manufacturers and time to market authorisation
Goal 2 Increase collaboration and information sharing with overseas comparable regulators to reduce regulatory burden
We will collaborate with international partners on information sharing, work sharing and sharing data to facilitate internationally consistent decisions about therapeutics products.
Information sharing, work sharing and sharing data between international regulators enables the TGA to make informed and internationally consistent decisions about therapeutic products. Working with comparable international regulators ensures products marketed in multiple countries meet the safety, quality and performance requirements of a globally aligned regulatory framework.
International collaboration on the evaluation of therapeutic products has the potential to reduce market authorisation timeframes. Sharing scientific resources and expertise and reducing duplication in the evaluation of products reduces costs and creates efficiencies to facilitate faster patient access to quality, safe and effective therapeutics. International collaboration extends to inspections of manufacturing facilities on behalf of regulatory authorities in other countries and relies on an understanding of the regulatory systems and standards between collaborating regulators, with final decisions made by the individual regulators.
Summary of Activities
- Continue being a WHO Essential Regulatory Laboratory and working with the Global Influenza Surveillance and Response System
- Collaborate in evaluation of COVID-19 vaccines and therapeutics
- Collaborate on Oncology Submissions through Project Orbis
- Utilise comparable overseas regulator pathways for prescription and complementary medicines and medical devices
- Participate in Pharmaceutical Inspections Cooperation Scheme (PIC/S) and key initiatives associated with inspection reliance
- Work with IMDRF to develop a risk calibrated regulatory approach for innovations and promote harmonised pre-market review requirements
- Increase information sharing and work sharing with overseas regulators to enhance scientific and technical expertise and reduce duplication in the evaluation of products through ACCESS
- Conduct domestic and international manufacturing inspections using digital technologies and share inspection information with other regulators
Goal 3 Utilise international networks to monitor product safety and quality and maintain supply chains
We will collaborate with international regulatory networks to identify post market issues, enhance monitoring systems and build a greater body of real-world evidence for informing regulatory decisions.
As therapeutic goods become more complex and pressure to reduce market authorisation timeframes increases, the need for effective global post market monitoring systems becomes paramount. International regulatory networks help identify post-market issues and align public communications to promote confidence in the use of products approved by regulatory authorities. Continued use of these networks and learning from other regulators about their activities will help build a greater body of real-world evidence to inform regulatory decisions.
The TGA plays an important role in facilitating, monitoring and enforcing compliance with the regulatory requirements for therapeutic goods in Australia. The growing online availability of health products increases the threat of unregulated or counterfeit products being supplied into Australia. Our networks with international regulators, agencies, and sponsors of products helps identify the illegal import and supply of goods, for the protection of consumers and a level playing field for Australian industry.
Manufacture of medicines and medical devices predominantly occurs offshore, using raw materials or components from a complex chain of supply. Environmental, geographical and political factors can interrupt supply, as can public health emergencies. Monitoring supply chains is critical to ensure mitigation strategies can be put in place early if difficulties maintaining supply emerge.
Summary of Activities
- Work with international pharmacovigilance and adverse event reporting programs using secure channels and confidentiality agreements to share reports if signals emerge
- Participate in relevant international forums
- Implement medical devices post market surveillance strategies
- Work with the Global Regulatory Drug Shortage working group to share information on efforts to prevent, monitor, and address drug shortages
- Permanent Forum on International Pharmaceutical Crime (PFIPC) to collaborate on information and intelligence sharing for the identification and assessment of emerging threats and trends, and to develop training that supports best practice investigations and intelligence capability
- Enhance regulatory science skills and capabilities of staff to ensure the TGA is prepared for advances in technology and emerging regulatory challenges
- Implement internationally aligned post market regulatory controls which complement pre-market requirements and manage evolving risks effectively
Goal 4 Strengthen regional regulatory capabilities for safer and effective therapeutics
We participate in technical assistance programs in the Asia Pacific Region to strengthen regulatory capability.
The TGA actively participates, and in some cases leads, in technical assistance programs across participating countries which span the WHO regional boundaries for the South-East Asia and the Western Pacific regions. National regulators across these areas are developing capabilities in product review, manufacturing assessment and pharmacovigilance. Improved regulatory practice reduces delays in accessing new and priority medicines and medical devices and helps identify substandard and/or falsified products. The TGA's work in the region will help national disease control efforts against tuberculosis and malaria, resulting in reduced health security threats to neighbouring countries and regions.
As part of Australia's regional response to the COVID-19 pandemic, the TGA will continue to expand its support to assist regional regulators with their response to the pandemic including preparedness for potential treatments and/or vaccines as they become available.
Summary of Activities
- Strengthen regulatory capacity across the South-East Asia and Pacific region through continued engagement with the Department of Foreign Affairs and Trade (DFAT)
- Focus on the COVID-19 pandemic response by working closely with the Australian DFAT Centre for Health Security to ensure support is reaching those in need in an effective and timely manner.
- 18 countries in South-East Asia and the Pacific are part of this program
- Provide ongoing COVID-19 pandemic response support across the region
- Continue to participate in the Indo Pacific Regulatory Strengthening Program
- Partner countries include Cambodia, Lao PDR, Myanmar, Vietnam, Indonesia, Papua New Guinea and Thailand
- Continue to work with DFAT on the Pacific Medicines Testing Program to provide Pacific Island counties access to Australian laboratory testing for medicines quality assurance and problem and complaint investigation.
- Countries include: Federated States of Micronesia, Fiji, Kiribati, the Marshall Islands, Nauru, Palau, Papua New Guinea, Samoa, Solomon Islands, Timor Leste, Tonga, Tuvalu, and Vanuatu
- Continue cooperation and engagement with the WHO to ensure our work aligns with broader regional public health priorities
- Continue to engage with and support WHO programs through the WHO designated Collaborating Centres for Drug Quality Assurance and Quality Assurance of Vaccines and other Biologicals
Major international forums
We will continue to participate in a range of international forums to support collaboration and align regulatory processes. The major forums that we participate in include:
- The International Coalition of Medicines Regulatory Authorities (ICMRA)
- The Centre for Innovation in Regulatory Science (CIRS)
- The International Medical Device Regulators Forum (IMDRF)
- Medical Device Single Audit Program (MDSAP)
- The International Council for the Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH)
- International Pharmaceutical Regulators Programme (IPRP)
- Pharmaceutical Inspections Cooperation Scheme (PIC/S)
- ACCESS - Australia, Canada, Singapore, Switzerland and the United Kingdom Consortium
- WHO - World Health Organization
Mechanisms to facilitate international engagement
An appropriate legal framework and enhancing our staff opportunities and skills to engage in international work underpins TGA's international engagement activities. Collaboration skills and the ability to contribute to strategic discussions are key indicators for the success of our engagements.
Scientists, clinicians and specialist advisers working on ensuring the safety, efficacy and quality of therapeutic products all benefit from having strong administrative support that allows them to focus on their areas or expertise. Provision of secretariat support for staff engaged in international work, facilitating meetings across global time zones and ensuring secure storage and access to confidential information is essential.
Establishment of legal agreements and treaties with international regulatory counterparts needs to also balance Australia's trade interests. These agreements ensure Australia's access to quality, safe and effective therapeutic goods is maintained when sharing confidential information and negotiating trade agreements. These include:
- Mutual Recognition Agreements - treaty level agreements to provide for the mutual recognition of specific activities undertaken by other regulators, e.g. manufacturing inspections
- Memoranda of Understanding - to support confidential information sharing and increase the range of national regulatory authorities that the TGA can use as Comparable Overseas Regulators
- Free Trade Agreements - are reviewed in collaboration with DFAT, with Australia's interests in relation to pharmaceuticals, intellectual property and data exclusivity matters often a key component to be negotiated e.g. with the EU, the UK, Regional Comprehensive Economic Partnership or the Transpacific Partnership
- International treaties - collaborating across Government to ensure Australia's interests are maintained. The implementation of international treaties may impact therapeutic goods regulations, for example, the Minamata Convention on Mercury
- Technical Barriers to Trade - are considered to ensure trade is not adversely impacted.

