When is it suitable to use a LBS?
The acceptance and evaluation of a LBS is considered by the TGA on a case-by-case basis according to the regulatory and clinical history of the medicine, both in Australia and overseas (especially in the UK, USA, Sweden, Canada, the Netherlands, and New Zealand).
The following table provides a general guide as to the application type that may be submitted for a given marketing history of a medicine:
| Years medicine marketed | Application type | ||
|---|---|---|---|
| Conventional | Mixed | LBS | |
| <5 yrs | 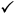 | 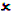 | 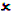 |
| 5-10 yrs | 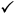 | 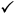 | 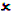 |
| >10 yrs | 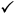 | 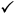 |  |
When is it not suitable to use a LBS?
Applicants should not routinely submit literature based data sets where sufficient company study reports are available. If the set of data generated from company study reports is incomplete, applicants may supplement the data with literature-based data.
LBS are generally not appropriate for applications to change the clinical trials section of a medicine's Product Information (PI). Applications to change this section usually require the submission of full study reports of the relevant clinical trials.
Literature-based data sets are not suitable for the quality (Module 3) component of an application as published reports rarely include sufficient validation information. A conventional Module 3 dossier should be submitted for all applications with a quality component.
LBS for new chemical entity (NCE) applications
Under exceptional circumstances, a LBS (including a mixed application) may be accepted for the registration of a new chemical entity (NCE) in Australia where it has been marketed in other countries for many years.
The quality (Module 3) component must be a conventional dossier.
We will not accept a LBS (or a mixed application) for applications to register a NCE where the marketing in other acceptable countries has been less than ten years, except where:
- the NCE has been designated in Australia as an Orphan Drug, or
- there is no medicine registered and available in Australia that is registered for the same or (in the view of the TGA) essentially the same indication.
Countries currently identified as acceptable are:
- Canada
- Sweden
- Netherlands
- United Kingdom
- United States of America
- New Zealand.
Types of LBS accepted
The dossier requirements for a LBS depend upon whether or not the application requires a systematic search of the literature.
The choice of LBS type will need to be assessed on a case by case basis.
For prescription medicines, applicants are encouraged to first discuss the details with the relevant clinical unit in OMA.
A general guide to the LBS type suitable for different types of applications is given below.
| Type of LBS | Type of application |
|---|---|
Based on systematic search of the literature Requires a fully reported systematic search of the literature as part of Module 1. |
|
Not generally requiring systematic search of the literature Does not require an extensive search of the literature, and reporting of the literature review forms part of the expert report(s). |
|
Literature based submissions for prescription medicines based on a systematic search of the literature
Pre-submission planning
The success (or otherwise) of any given application is based on the quality of the relevant data submitted in support of that application, and whether efficacy and safety is considered by the TGA to have been adequately demonstrated. A LBS is no different.
You should consider the following before discussing the proposed LBS with us:
- the aims of the application - for example, changes to the PI
- the nature of the application - LBS or mixed application
- the supporting data requirements - CTD Module 4 (nonclinical) and/or CTD Module 5 (clinical) data
- the appropriate literature search methodology.
You should plan to discuss these and any other issues with us at least three months prior to the pre-submission phase (for further information, see Consultation with TGA).
Consultation with TGA
Before lodging your Pre-submission planning form (PPF), you need to consult with us to ensure that the proposed approach to a LBS is appropriate to both the medicine and the application type.
How to commence consultation for a LBS
The TGA point of contact is the OMA Case Manager of the relevant prescription medicine clinical unit. Information on the areas of responsibility for the clinical units is included in Prescription medicine clinical units.
Initial consultation can be via email, and several rounds of discussion may be necessary.
If you would like to arrange a formal pre-submission meeting with us, follow the guidance within Pre-submission meetings with the TGA.
Applications that do not require a systematic review of the literature can usually be handled through correspondence; however formal meetings can be arranged for these if you wish.
What to do prior to the pre-submission meeting
To ensure that the discussion can link relevant aspects of the application to the concepts to be addressed in the literature search, provide the following information 2 weeks prior to the meeting (in addition to the documentation described in the guidance on pre-submission meetings):
- a discussion paper that outlines the scope of the application and issues to be addressed at the meeting
- a draft literature search strategy, including proposed database search strategies, a list of proposed databases and/or other sources to be used
- a justification for the approach taken
- the criteria for determining which search results are to be included/excluded in the application dossier.
What you can expect to happen
We will appraise proposed literature searches and attend pre-submission meetings.
We will provide feedback on the proposed methodology and, if changes are required, the literature search strategy must be amended and resubmitted.
It is not sufficient to assert that any required changes will be made, so ensure you do not finalise your application dossier until we have approved your search strategy.
TGA approval of the search strategy
If we agree that a LBS is appropriate, you should discuss, and seek our agreement, on the following before the LBS is compiled:
- search requirements (systematic or non-systematic) and methodology to be used
- criteria for determining which papers identified by the search results are to be included/excluded from the application dossier.
The literature search strategy should be amended and resubmitted until we have notified you that it is acceptable, so ensure you do not submit a PPF until you have received this notification.
Note: Do not finalise the dossier for a literature based submission until your search strategy has been approved, as any changes may have flow-on effects for revisions to other modules.
Please also be aware that:
- Agreement on the strategy to be used in the literature search does not guarantee the success of the LBS.
- The alteration of a search strategy, or inclusion/exclusion criteria, after we have agreed to them, is not acceptable.
- The responsibility for the choice of papers from the search output for inclusion in the application rests with the sponsor.
- There must be no unjustified discrepancy between the search output and the set of evidence to be scrutinised. The dataset retrieved by the approved literature search should be the same as that provided in the application.
- Notwithstanding the pre-approval of the search strategy, we may subsequently raise questions during the evaluation process regarding the evidence base and the selection of papers submitted.
Once we have agreed to the proposed search strategy
Prior to lodging the PPF for a LBS, we will need to have reviewed and provided written agreement for:
- the literature search strategy, including the databases to be searched
- the criteria for determining which search results are to be included/excluded in the application dossier.
Ensure a copy of our agreement to the search strategy is included as an attachment to the PPF.
If a LBS application is delayed
If substantial time has elapsed between our agreement to the search strategy and the proposed submission of the dossier, you may need to:
- update the literature search and revise the dossier to take account of new evidence
- justify the continued use of an earlier search in these cases.
When determining whether a search is up to date, we will take into account a reasonable amount of time for the application to be compiled (not more than 6 months before lodging the PPF).
Note: Inform the TGA Delegate immediately if you become aware of any significant new information that has arisen after the search date.
Other sources of information on how to prepare a LBS
- National Health and Medical Research Council (NHMRC) handbook: How to review the evidence: systematic identification and review of the scientific literature.
Advice may be sought from the TGA on the literature searches required for the application via the OMA Case Manager.
Literature based submissions not based on a systematic search of the literature
Requirements
This LBS type does not need to be based on an extensive search of the literature.
There is no need to provide copies of a search strategy and output; however, ensure the dossier contains an overview(s) that:
- justifies the choice of a non-systematic review of the literature, rather than a systematic review
- addresses the proposed changes in the context of an appraisal of all the submitted papers
- addresses the proposed changes in the context of any previously submitted data.
Changes to existing PI documents based on previously submitted data
It is particularly important that you justify the relevance of the new data in relation to the old data.
Examples of the type of changes that require this include:
- deletion of a precaution or effects on laboratory tests.
Changes to existing PI documents for special patient groups or based solely on nonclinical data
Include discussion and justification when:
- changes to the PI that modify the wording for use of the medicine in special patient groups (e.g. lactating patients or those with renal or hepatic impairment)
- changes are based solely on nonclinical data (e.g. insertion of positive carcinogenicity or genotoxicity results from nonclinical studies).
Use of a medicine in pregnancy
Information on the use of any medicine in pregnancy should generally be worded in accordance with the Prescribing medicines in pregnancy database.
If you wish to use an alternative statement, your justification should be in the context of:
- the supporting data
- any data previously submitted.
General advice
To aid in the presentation and evaluation of this type of application, ensure that:
- the data are submitted according to the CTD guidelines
- all submitted papers are cross-referenced in the Module 2 summaries and overviews so the papers can be located easily
- where the proposed changes have been approved in an overseas countries, the basis on which such an approval was given is described
- for very old medicines - where evidence from the literature is not available to be submitted:
- For prescription medicines, the use of standard texts may be appropriate, the use of reference textbooks and, particularly, what constitutes an appropriate reference textbook, should be discussed with the OMA Delegate at the earliest opportunity.
- For OTC medicines, refer to Australian Regulatory Guidelines for OTC Medicines (ARGOM), Safety and efficacy data, Section 4.1 Literature-based submissions.

