Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
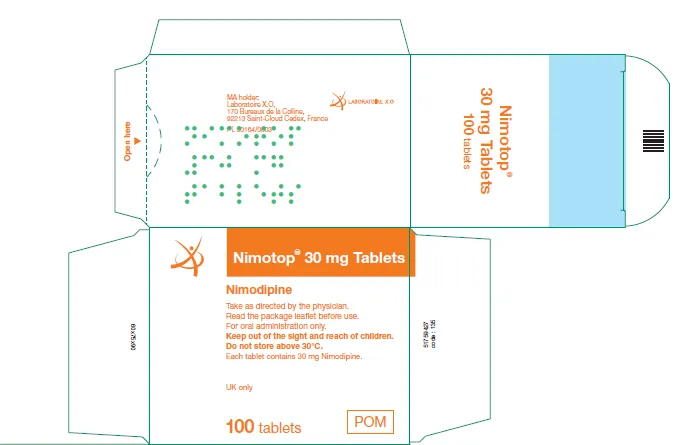
(Approval lapsed) NIMOTOP nimodipine 30mg tablets (UK)
Section 19A approved medicine
(Approval lapsed) NIMOTOP nimodipine 30mg tablets (UK)
Section 19A approval holder
Boucher & Muir Pty Ltd T/A Advanz Pharma ABN 58 000 140 474
Phone
1800 627 680
Approved until
Status
Expired
Medicines in short supply/unavailable
NIMOTOP nimodipine 30 mg tablets blister pack - ARTG 43100
Indication(s)
Prophylaxis of ischaemic neurological deficits caused by cerebral vasospasm after subarachnoid haemorrhage following ruptured intracranial aneurysm
Images