Medical Devices Safety Update is the medical device safety bulletin of the Therapeutic Goods Administration (TGA)
How the TGA uses Australian joint replacement registry data
The information generated by the Australian Orthopaedics Association National Joint Replacement Registry has been an important tool for post-market vigilance and monitoring and for decisions regarding inclusion of medical devices on the Australian Register of Therapeutic Goods (ARTG).
The Australian Orthopaedics Association National Joint Replacement Registry (AOANJRR) has been operating since 1999. Every October the registry publishes an annual report for the previous year, alongside several supplementary reports. The 2017 annual report undertook analysis on 1,237,576 joint replacement procedures (545,831 hip, 653,480 knee and 38,265 shoulder). Registry information is also available through a web portal.
The annual reports contain information about the types and reasons for revision procedures and identify implant combinations that have higher-than-anticipated rates of revision. The information is a resource for orthopaedic surgeons, implant manufacturers, researchers and regulatory agencies such as the TGA.
A joint replacement procedure triggers data collection including: patient identifying details; consulting surgeon; reason for surgery; and the type of implant, including individual implant components. If the procedure is a revision the type of revision and reason for revision are recorded. Although patient identifiers are collected, they are only used to ensure the integrity of the data and to reconcile revision information with the original operation. In case of a problem, it is best for the patient to be contacted by a medical practitioner.
Why is it so useful?
The AOANJRR reports are useful for a variety of reasons including:
- The 'opt-out' method of enrolment leads to a virtually complete data set.
- A wide variety of analysis options are available.
- The data set allows calculation of Cumulative Percent Revision (Kaplan-Meier survivorship).
- Revision rates can be broken down among particular patient populations, for example: primary diagnoses; gender; age; use of cement; type of implant; implant models; etc.
- The reasons for the primary procedures and types of revision procedures can be taken into account.
Focus on outliers
The TGA's focus has been on implants that are revision rate outliers. An implant may be having a higher-than-expected rate of revision for many reasons. These must be considered before contemplating regulatory action.
In 2006 the TGA established a process for the investigation of an implant 'identified' as having a higher-than-expected rate of revision:
- The sponsor/manufacturer of the implant is requested to make a submission relating to the safety and performance of the implant.
- A group of orthopaedic experts considers the information from the AOANJRR and the manufacturer. The expert group advises the TGA about safety, performance and benefits that may compensate for the high risk of revision.
- The TGA considers all the evidence and the expert advice and makes a regulatory decision. The action taken could be a recall, hazard alert, safety alert and/or cancellation, or taking no action if it is appropriate.
In 2017 the registry 'identified' 130 implant combinations:
- All 130 implant combinations have been investigated by the TGA.
- 53 implant combinations are in the category 'identified and still used'
- 77 implant combinations are in the category 'identified and no longer used'
- 32 of these 77 implant combinations were withdrawn from the market after some form of TGA intervention.
TGA intervention is not restricted to recall or product cancellation. The TGA has also intervened in instances where the implant continues to be used, for instance by ensuring that clear advice is provided about the use indications and contraindications of particular implants.
The outcomes of the interventions are published on the TGA website (www.tga.gov.au). The Australian Orthopaedic Association and surgeons who used the implants that are subject to regulatory outcomes are notified individually by the sponsor of the implant.
Other uses
The TGA also uses registry information to check the revision rate of implants that are the subject of a Device Incident Report(An adverse event report to the TGA regarding a medical device).
We have also used registry information in the assessment of applications for new implants. In some cases, the TGA has used AOANJRR reports on clinically equivalent implants to compensate for a lack of sufficient clinical evidence about a new device.
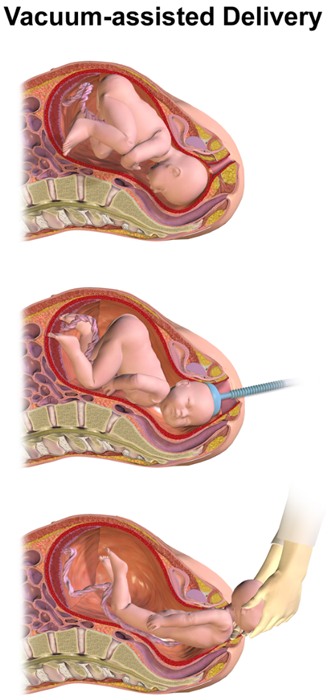
Focus on skills for vacuum-assisted births
The safe use of devices in vacuum-assisted births relies on careful patient selection, good technique, and the setting of appropriate procedural limits, all within a robust clinical governance framework.
There is a reported increase in clinical acceptance and use of medical devices in vacuum-assisted deliveries and so far the TGA has not received any serious injury reports related to the use of these devices. However, device sponsors and clinical experts have been calling to get more attention to appropriate training, supervision and credentialing as vacuum-assisted deliveries, like all similar procedures, are not free of potentially serious risks.
NSW's Clinical Excellence Commission (CEC) addressed many of the issues in a 2014 report, Vacuum Assisted Births - Are We Getting it Right? A focus on subgaleal haemorrhage.[1] The report found vacuum delivery was not without risk with injuries in about 5 per cent of deliveries. Complications include subgaleal haemorrhage, which is potentially life threatening and occurs in approximately 1 in 300 cases.[2]
CC BY-SA 4.0 (//creativecommons.org/licenses/by-sa/4.0),
via Wikimedia Commons.
After analysing data collected by NSW Health's Incident Information Management System, the CEC report concluded:
Management of the second stage of labour can be challenging. With respect to vacuum assisted births, clinicians need to appreciate that while the incidence of maternal trauma is reduced compared with forceps, neonatal trauma occurs in approximately 1 in 15 babies. While such trauma is mostly minor, potentially fatal complications such as subgaleal haemorrhage do occur.
It is evident from the review of cases that clinicians may perceive that the use of vacuum devices do not require the same level of rigour with respect to training, supervision and credentialing, as other forms of assisted birth. The prerequisites for instrumental vaginal birth need to be fulfilled and documented. There must be adequate maternal effort such that a clinician needs to question the use of vacuum devices where there is profound maternal exhaustion or where a neuraxial block (e.g. epidural) significantly inhibits the mother's expulsive efforts. Vacuum devices should not be used for births less than 36+0 weeks gestation and never before 34+0 weeks gestation. However, if a vacuum assisted birth is to be performed at 36+0 weeks gestation a consultant should be present to provide direct supervision. Professionally determined procedural limits must be adhered to and documented.
Many cases reviewed in this report indicate that clinician supervision, skill and knowledge are variable across the system. In particular, it would appear that some clinicians do not possess the full range of obstetric skills that would permit alternative options to effect birth safely. Professional obstetrical and gynaecological bodies recognise the need for clinicians to be skilled in both forceps and vacuum assisted births early in their career development. Such skills require appropriate training, supervision and credentialing.
Instrumental vaginal birth continues to have a role in modern obstetrics. It is recognised that forceps births are associated with an increase in maternal trauma. However, this report would indicate that the risk of neonatal trauma in vacuum assisted births is not fully appreciated. It is important for clinicians to recognise those elements of safe instrumental vaginal birth that are critical to minimising harm to both mothers and babies.
These conclusions echo similar findings in a 2010 Cochrane review, Instruments for assisted vaginal delivery:[3]
There is a recognised place for forceps and all types of ventouse in clinical practice. The role of operator training with any choice of instrument must be emphasised. The increasing risks of failed delivery with the chosen instrument from forceps to metal cup to hand-held to soft cup vacuum, and trade-offs between risks of maternal and neonatal trauma identified in this review need to be considered when choosing an instrument.
References
- Vacuum Assisted Births - Are We Getting it Right? A focus on subgaleal haemorrhage. Clinical Excellence Commission 2014. http://www.cec.health.nsw.gov.au/__data/assets/pdf_file/0010/258247/c-f-report-vacuum-assisted-births-are-we-getting-in-right.pdf (accessed 10 May 2018)
- Prevention, Detection and Management of Subgaleal Haemorrhage in the Newborn. The Royal Australian and New Zealand College of Obstetricians and Gynaecologists. 2015; C-Obs 28. https://www.ranzcog.edu.au/RANZCOG_SITE/media/RANZCOG-MEDIA/Women's%20Health/Statement%20and%20guidelines/Clinical-Obstetrics/Prevention-detection-and-management-of-Subgaleal-Haemorrhage-(C-Obs-28)-Review-November-2015.pdf?ext=.pdf (accessed 10 May 2018)
- Instruments for assisted vaginal delivery. O'Mahony F, Hofmeyr GJ, Menon V. www.cochrane.org/CD005455/PREG_instruments-for-assisted-vaginal-delivery (accessed 10 May 2018)
TGA reviews product safety of ventilators
The TGA is undertaking a product safety review into ventilators being used in the intensive care or high-level care environments.
The TGA has requested that sponsors of ventilators provide the following information:
- evidence that the ventilators meet the ventilation delivery and monitoring specifications published in the Instructions For Use, which should include evidence that the breathing circuits recommended for use with this ventilator are valid
- for ventilators that are intended for use with paediatric patients, a clear clinical justification and validation of why tidal volume delivery specifications are acceptable and safe
- post-market data
- which breathing circuits are suitable for use with the ventilator.
The information requested is undergoing review and if any regulatory actions resulting from the review are taken, it will be published.
More information about the review and the products included is available on the TGA website.
What to report? Please report adverse events, as well as near misses
The TGA encourages the reporting of any suspected adverse event or potential adverse event relating to a medical device. Adverse events can involve actual harm to a patient or caregiver, or a near miss that may have resulted in harm.
Some issues relating to medical devices that may lead to adverse events and prompt you to report include:
- mechanical or material failure
- design issues
- labelling, packaging or manufacturing deficiencies
- software deficiencies
- device interactions
- user/systemic errors.
Suspected adverse events or near misses can be reported directly to the TGA:
- online at Report a problem
- by emailing iris@tga.gov.au
- by mail to IRIS, TGA, PO Box 100, Woden ACT 2606
- by fax to 02 6203 1713.
For more information about reporting, visit www.tga.gov.au or contact the TGA's Medical Devices Branch on 1800 809 361.
Disclaimer
The Medical Devices Safety Update (MDSU) is aimed at health professionals and is intended to provide practical information on medical device safety, including emerging safety issues. The information in the MDSU is necessarily general and is not intended to be a substitute for a health professional's judgment in each case, taking into account the individual circumstances of their patients. Reasonable care has been taken to ensure that the information is accurate and complete at the time of publication. The Therapeutic Goods Administration gives no warranty that the information in this document is accurate or complete, and does not accept liability for any injury, loss or damage whatsoever, due to negligence or otherwise, arising from the use of or reliance on the information provided in this document.
© Commonwealth of Australia 2018
This work is copyright. You may reproduce the whole or part of this work in unaltered form for your own personal use or, if you are part of an organisation, for internal use within your organisation, but only if you or your organisation do not use the reproduction for any commercial purpose and retain this copyright notice and all disclaimer notices as part of that reproduction. Apart from rights to use as permitted by the Copyright Act 1968 or allowed by this copyright notice, all other rights are reserved and you are not allowed to reproduce the whole or any part of this work in any way (electronic or otherwise) without first being given specific written permission from the Commonwealth to do so. Requests and inquiries concerning reproduction and rights are to be sent to the TGA Copyright Officer, Therapeutic Goods Administration, PO Box 100, Woden ACT 2606 or emailed to tga.copyright@tga.gov.au.
For the latest information from the TGA, subscribe to the TGA Safety Information email list.
For correspondence or further information about Medical Devices Safety Update, contact the TGA's Medical Devices Branch at iris@tga.gov.au or 1800 809 361.
Medical Devices Safety Update is written by staff from the Medical Devices Branch.
Editor: Ms Pamela Carter
Deputy Editor: Mr Aaron Hall
TGA Chief Medical Adviser: Adjunct Professor Tim Greenaway
Contributors include: Ms Jane Shum , Dr Amanda Craig and Dr Jorge Garcia.
Print version
Related content
-
Orthopaedic implant revision rate data
Media releasesThe annual report contains information about hip, knee and shoulder orthopaedic implants that are having higher-than-expected rates of revision. -
Medical Devices Safety Update, Volume 6, Number 1, January 2018
Safety updatesInappropriate use of scalpel removers; ransomware named as 2018's 'top hazard'; joint registry data; recent safety alerts -
Medical Devices Safety Update, Volume 3, Number 4, July 2015
Safety updatesIn this issue: Action after registry data analysis; Environmental extremes; Button battery dangers; Infant sleep positioners reviewed

