Consumers and health professionals are advised that Medtronic Australasia, in consultation with the TGA, has issued a hazard alert regarding multiple models of its spinal cord stimulation devices.
Medtronic Australasia is also undertaking a recall for product correction to update the Instructions for Use (IFU).
Spinal cord stimulation devices are implantable, programmable medical devices that deliver electrical stimulation to the patient's spinal cord.
Spinal cord stimulation devices are used to help treat certain types of chronic pain in the torso and/or limbs, and pain associated with certain vascular and heart conditions.
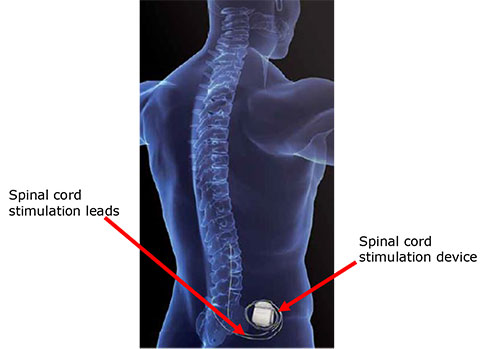
It has been identified that patients who have received Medtronic spinal cord stimulation devices (see below for lists of devices affected by the hazard alert, including neurostimulators, leads, kits and programmers) have a very small risk of developing a tissue mass around a device lead that can cause spinal cord compression.
The estimated rate of occurrence of this problem globally is less than 1 in 10,000 patients. In reported cases, the onset of symptoms ranged from weeks to 17 years after the device was implanted.
If spinal cord compression occurs, the patient may experience loss of pain relief and neurological symptoms, such as muscle weakness in their limbs.
Surgery may be required to remove a mass causing spinal cord compression.
To address this issue, Medtronic Australasia is updating the IFUs for the affected devices to include specific information regarding the potential for a mass to form around a device lead, which could lead to spinal cord compression.
Awareness of this potential adverse event can lead to early detection and prevention of associated adverse events.
Information for consumers
Problems associated with this issue are rare and removal of implanted devices in the absence of symptoms is not recommended.
Medtronic Australasia has written to health professionals who have implanted spinal cord stimulation models affected by the hazard alert, or are managing patients who have these devices, providing further information.
If you have a spinal cord stimulation device implanted and have any questions or concerns regarding this issue, contact your managing health professional.
Information for all health professionals
If you are treating a patient who has a spinal cord stimulation device implanted, reassure them that problems associated with this issue are rare.
If a patient has any questions or concerns about this issue, refer them to their managing surgeon or neurologist.
Information for surgeons and neurologists
Medtronic Australasia has written to surgeons and neurologists who have implanted spinal cord stimulation models affected by the hazard alert, or are managing patients who have these devices, providing further information.
At their next scheduled visit, advise patients of this issue and reassure them that problems associated with it are rare.
Prophylactic removal of implanted Medtronic spinal cord stimulation leads is not recommended.
If a patient presents with a new neurological deficit, you should consider this issue as a potential cause.
If an asymptomatic epidural mass is identified, consider periodic monitoring.
Medtronic has identified 14 reports over the last 30 years worldwide describing delayed spinal cord compression due to an epidural mass consisting of reactive tissue (granulomatous and/or fibrous tissue) around a Medtronic spinal cord stimulation lead. All patients had neurological deficits and required surgery to remove the mass. The severity of symptoms ranged from muscle weakness to progressive quadriparesis. The onset of neurological symptoms was often preceded by a loss of therapy and, in several cases, only one side of the body was affected.
If you have any questions or concerns regarding this issue, contact Medtronic Australasia customer service on 1800 668 670.
Affected devices
Below are lists of spinal cord stimulation devices, leads, kits and patient programmers for which the adverse event labelling will be updated.
Spinal cord stimulation devices and model numbers:
- Synergy - 7427
- Synergy Versitrel - 7427V
- Prime Advanced - 37702
- Restore - 37711
- RestoreUltra - 37712
- RestoreAdvanced - 37713
- RestoreSensor - 37714
- PrimeAdvanced SureScan MRI - 97702
- RestoreUltra SureScan MRI - 97712
- RestoreAdvanced SureScan MRI - 97713
- RestoreSensor SureScan MRI - 97714.
Spinal cord stimulation leads and model numbers:
- Pisces Quad - 3487A
- Resume II - 3587A
- 1x8 Sub-Compact - 3776
- 1x8 Standard - 3777
- 1x8 Compact - 3778
- 1x8 Sub-Compact Lead with Percutaneous Extension - 3876
- 1x8 Standard Lead with Percutaneous Extension - 3877
- 1x8 Compact Lead with Percutaneous Extension - 3878
- 1x8 Subcompact SureScan MRI - 977A1
- 1x8 Compact SureScan MRI - 977A2
- Pisces Quad Compact - 3887
- Pisces Quad Plus - 3888
- Resume TL - 3986A
- Specify - 3998
- Specify 2x4 Hinged - 3999
- Specify 2x8 - 39286
- Specify 5-6-5 - 39565
- 1x8 Subcompact SureScan MRI Trialling - 977D160
- 1x8 Compact SureScan MRI Trialling - 977D260.
Kits and model numbers:
- Lead Revision Kit - 3550-22
- Lead Revision Kit - 3550-23.
Patient programmers and model numbers:
- Itrel EZ Patient Programmer - 7434A
- Synergy Patient Programmer - 7435
- Patient Programmer - 37744
- Patient Programmer MRI SureScan - 97740.
Reporting problems
Consumers and health professionals are encouraged to report problems with medical devices. Your report will contribute to the TGA's monitoring of these products. For more information see the TGA Incident Reporting and Investigation Scheme (IRIS).
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medical device.

