Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Foreword
The Therapeutic Goods Administration (TGA) is responsible for regulating medicines, vaccines and biologicals, including monitoring the ongoing safety, quality and efficacy of these products once they have been included on the Australian Register of Therapeutic Goods (ARTG) through product vigilance activities.
On an annual basis, the TGA's Pharmacovigilance and Special Access Branch (PSAB) prepares a report for incorporation into the Department of Health publication Australian Statistics on Medicines.
Australian Statistics on Medicines is produced by the Drug Utilisation Sub-Committee of the Pharmaceutical Benefits Advisory Committee and is aimed at providing comprehensive and valid statistics on the Australian use of medicines and vaccines in the public domain to allow access by all interested parties.
This report from the PSAB includes a brief overview on the following aspects of post-market monitoring of medicines and vaccines in Australia:
- Adverse event reporting statistics for 2017
- Processing and use of adverse event reports
- Database of Adverse Event Notifications
- Reporting adverse events
- Expert advisory committees
- Medicines Safety Update
- Product vigilance
Adverse event reporting statistics for 2017
The TGA's reporting system for adverse events began in the late 1960s with the computerised database dating back to the early 1970s. By the end of 2017 there were approximately 348,000 reports of suspected adverse events in the database.
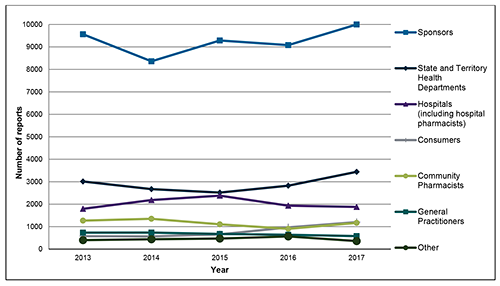
Figure 1: Origin of medicine and vaccine adverse events received by the TGA (2013-17)
| Source | 2013 | 2014 | 2015 | 2016 | 2017 |
|---|---|---|---|---|---|
| General Practitioners | 733 | 735 | 675 | 639 | 579 |
| Hospitals (including hospital pharmacists) | 1794 | 2184 | 2381 | 1934 | 1879 |
| Community Pharmacists | 1268 | 1351 | 1105 | 903 | 1170 |
| Sponsors | 9563 | 8359 | 9286 | 9080 | 9998 |
| Consumers | 574 | 565 | 654 | 969 | 1201 |
| State and Territory Health Departments | 3011 | 2672 | 2516 | 2824 | 3441 |
| Other | 400 | 441 | 470 | 567 | 359 |
| Total | 17343 | 16307 | 17087 | 16916 | 18627 |
In 2017 the TGA received approximately 18,600 reports of adverse events. As shown in Figure 1, the majority of reports made in 2017 were by sponsors. The number of reports made by sponsors decreased from its previous high in 2013 (9563) to its lowest point for this five-year period in 2014 (8359), before eventually increasing to a new high in 2017 (9998).
Of the 18,600 total adverse event reports received by the TGA in 2017, approximately 54% (9998) were from sponsors; 18% (3441) from State and Territory Health Departments (reports of adverse events following immunisation); 10% (1879) from hospitals and hospital pharmacists; 7% (1201) from consumers; 6% (1170) from community pharmacists; 3% (579) from general practitioners (GPs); and 2% (359) from other sources.
Compared to previous years, the biggest increases in reports were from sponsors (up from 9080 in 2016 to 9998 in 2017) and State and Territory Health Departments (up from 2824 in 2016 to 3441 in 2017).
The increase in reports from State and Territory Health Departments may relate, in part, to the continuation of enhanced surveillance for two additions to the National Immunisation Program in 2016 - the 18-month diphtheria, tetanus and acellular pertussis-containing vaccine and Zostavax.
While health professionals are encouraged to report suspected adverse events directly to the TGA, they can also report to the sponsor or manufacturer.
Processing and use of adverse event reports
The PSAB assessed adverse event reports submitted to the TGA by checking for the presence of 'minimum' details, including an individual patient, an adverse event, at least one (suspected) medicine or vaccine, and an identifiable reporter. The specific adverse event terms are identified along with the suspected, interacting or 'other' therapeutic products and these are entered into the database.
The PSAB assesses causality of adverse event(s) and in some cases requests further clinical or laboratory information from the reporter. Medical officers review serious reports and branch staff regularly analyse reporting data to identify potential safety signals.
Reports are forwarded to the Uppsala Monitoring Centre in Sweden, which administers the World Health Organization Collaborating Centre for International Drug Monitoring. This global database began in 1968 as a pilot program involving 10 nations, including Australia, and now receives reports from more than 80 nations.
Database of Adverse Event Notifications
Information in the publicly searchable Database of Adverse Event Notifications (DAEN) comes from reports made to the TGA by a wide range of sources, including members of the public, general practitioners, nurses, other health professionals and the therapeutic goods industry. Reports in this database start from 1 January 1971 up to three months prior to the date of access. The TGA uses this three-month period to investigate any potential safety concerns identified.
The DAEN, which was launched in 2012, was created to support better health outcomes by providing access to the information that the TGA gathers while monitoring medicine and vaccine safety in Australia.
Reporting adverse events
The TGA encourages the reporting of all suspected adverse events to medicines and vaccines available in Australia, including prescription medicines, over the counter and complementary medicines. The reporting of seemingly insignificant or common adverse events can contribute to the TGA's investigation of a potential safety signal.
The TGA particularly encourages reporting of:
- suspected adverse events involving new medicines and vaccines
- suspected medicine and vaccine interactions
- unexplained adverse events (adverse events that are not described in the Product Information)
- serious adverse events, such as those suspected of causing:
- inability to work
- admission to hospital
- prolongation of hospitalisation
- increased investigation or treatment costs
- danger to life
- birth defects
- death.
For further information about reporting suspected adverse events, visit the TGA website (click on 'Reporting problems').
Sponsors of all medicines and vaccines on the ARTG have mandatory reporting requirements regarding adverse events.
Expert advisory committees
Advisory Committee on Medicines
The Advisory Committee on Medicines (ACM) was formed on 1 January 2017. The ACM provides independent medical and scientific advice to the Minister for Health and the TGA on issues relating to the safety, quality and efficacy of medicines supplied in Australia including issues relating to premarket and post-market functions for medicines.
The ACM is established under Regulation 35 of the Therapeutic Goods Regulations 1990 and the members are appointed by the Minister for Health. Membership comprises of professionals with specific scientific, medical or clinical expertise, as well as appropriate consumer health issues relating to medicines.
The ACM consolidated the previous functions of the Advisory Committee on the Safety of Medicines, the Advisory Committee on Prescription Medicines and the Advisory Committee on Non-Prescription Medicines.
Advisory Committee on Vaccines
The Advisory Committee on Vaccines (ACV) was formed on 1 January 2017. The ACV provides independent medical and scientific advice to the Minister for Health and the TGA on issues relating to the safety, quality and efficacy of vaccines supplied in Australia including issues relating to premarket assessment, post-market monitoring and safe use in national immunisation programs.
The ACV is established under Regulation 39F of the Therapeutic Goods Regulations 1990 and the members are appointed by the Minister for Health. Membership comprises professionals with expertise in specific scientific, medical or clinical fields, or consumer health issues.
The ACV consolidated the previous functions of the Advisory Committee on the Safety of Vaccines and the premarket functions for vaccines of the Advisory Committee on Prescription Medicines.
Medicines Safety Update
The Medicines Safety Update was published five times during 2017. It was published on the TGA website. Medicines Safety Update replaced the Australian Adverse Drug Reactions Bulletin in 2010.
The following articles were published in Medicines Safety Update during 2017:
- ibrutinib - ventricular tachyarrhythmia
- hepatitis B reactivation and infection
- improving Product Information
- codeine-containing products - use in children and ultra-rapid metabolisers
- lithium level monitoring and toxicity
- new precautions for hyoscine butylbromide ampoules
- off-label prescribing
- intravenous solution bags
- allergens
- medSEARCH app
- Viekira PAK and Viekira PAK-RBV - interaction with ethinyloestradiol
- ingenol mebutate - severe allergic reaction
- herpes zoster and eye injury
- testosterone and arterial thromboembolism/venous thromboembolism
- certain medicines and progressive multifocal leukoencephalopathy
- aripiprazole and impulse control disorders
- vemurafenib and risk of radiation injury.
Product vigilance
The TGA applies a risk management approach to ensure therapeutic goods supplied in Australia meet acceptable standards of quality, safety and efficacy. Once a therapeutic product is approved, the TGA continues to monitor the product in the market through therapeutic product vigilance activities.
The aim of therapeutic product vigilance is to continually monitor and evaluate the safety and efficacy (performance) profile of therapeutic goods and to manage any risks associated with individual products over their life cycle.
The maintenance and improvement of health and safety is a shared responsibility. In addition to government and industry, health professionals, consumers and their respective associations play an important role in reporting safety related issues.
Sponsors have the primary responsibility for the safety of any therapeutic products they import into, supply in or export from Australia. Sponsors must comply with legislative requirements for therapeutic product vigilance under the Therapeutic Goods Act 1989 (the Act) and there are applicable offences and penalties under the Act for not complying. The legislative requirements for therapeutic product vigilance vary depending on the type of therapeutic good.
The TGA commenced the Pharmacovigilance Inspection Program (PVIP) in September 2017 as part of its response to the Review of Medicine and Medical Device Regulation. The PVIP helps sponsors of medicines meet and maintain their pharmacovigilance obligations.
The TGA maintains up-to-date safety information on therapeutic products that is communicated through a variety of means to consumers and health professionals. The TGA is committed to advancing public health through market authorisation of beneficial, innovative therapeutic goods and by providing timely, evidence-based and authoritative information to allow consumers and health professionals to make informed decisions.
The TGA defines therapeutic product vigilance tools as tools designed to facilitate the collection and evaluation of information pertaining to the benefits and risks associated with the use of therapeutic products. The main product vigilance tools used by the PSAB are adverse event reports, RMPs and Periodic Safety Update Reports (PSURs).
Adverse event reports are reports of any unwanted and sometimes harmful occurrences from using medicines, vaccines or medical devices (collectively known as therapeutic goods). Importantly, adverse events related to the use of a therapeutic good are not always caused by the therapeutic good itself.
RMPs provide a summary of the known important safety information about the therapeutic product, plans to identify and characterise known or potential safety concerns and plans to minimise any identified or potential safety risk. A full outline of the scope of RMPs is above (see 'Expert advisory committee'). PSURs give an annual overview of the safety of the product, including adverse events, a summary of its registration status world-wide, actions taken for safety reasons, the world-wide usage of the product and an analysis of safety requirements. Sponsors must submit PSURs to the TGA for at least three years after registration of a product.
An important aspect of product vigilance is ensuring there are mechanisms to communicate safety information to both consumers and health professionals. To achieve this, the TGA publishes Australian Public Assessment Reports (AusPARs) about recently registered prescription medicines and vaccines on the TGA website. AusPARs outline the findings of the TGA's evaluation of a product including important effectiveness and safety information.
Each adverse event report the TGA receives is entered into a database, which is continually analysed by TGA staff to identify potential emerging problems for detailed investigation.
If the TGA identifies a safety concern relating to a medicine or vaccine, we can take regulatory action. This can include:
- informing health professionals and consumers via safety alerts, Early Warning System monitoring communications and Medicines Safety Update, which provide information and recommendations about therapeutic goods
- updating the Product Information, Consumer Medicine Information and/or product labelling with new adverse effects, precautions or warnings
- requiring post-marketing studies
- imposing limits on their use
- investigating manufacturing sites
- recalling products from the market
- suspending or cancelling products.
When a product is cancelled, details are published on the TGA website.
| Version | Description of change | Author | Effective date |
|---|---|---|---|
| V1.0 | Original publication | Pharmacovigilance and Special Access Branch | August 2018 |

