Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Andrographis paniculata (also known as green chireta) is a herb that is currently permitted for use in listed medicines as an active or homeopathic ingredient. Andrographis paniculata is typically used in relation to immune support and immune stimulating effects, such managing colds.
The TGA has previously reviewed the safety of Andrographis paniculata for use in listed medicines after receiving reports of adverse events associated with its use. The TGA published a safety review in 2015 that suggested an association between Andrographis paniculata and anaphylactic/allergic reactions and also published a safety alert.
Since the review in 2015, the TGA has continued to receive adverse event reports concerning anaphylactic/allergic reactions associated with medicines that contain Andrographis paniculata. This further supports a causal association between this ingredient and anaphylactic/allergic reactions. In light of this, the TGA considers that a new requirement for a warning statement on listed medicines is warranted to warn consumers about the risk of anaphylactic/allergic reactions with these medicines.
Given the nature of the risk to some consumers, all listed medicines released for supply from 2 May 2020 will need to comply with the new requirements outlined below.
Background
In 2015, the TGA completed a safety review of Andrographis paniculata and anaphylactic/allergic reactions that considered adverse events reported to the TGA as well as literature relating to the use of Andrographis paniculata. As part of the review, the TGA sought advice at the 23rd meeting of the Advisory Committee on the Safety of Medicines (ACSOM) on 11 July 2014. ACSOM advised that there was sufficient information to confirm an association between allergic/anaphylactic reactions and Andrographis paniculata. The committee also advised that the risk be communicated to the public, and that adverse event reports are monitored to assess the effectiveness of any risk mitigation strategy.
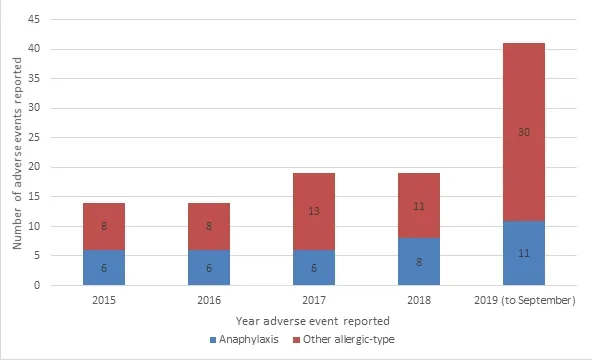
Since January 2015, the TGA has received a further 70 Australian reports of allergic reactions related to Andrographis paniculata and an additional 37 reports of anaphylaxis. For 11 of these cases, the sole suspected medicine contained Andrographis paniculata as the single active ingredient, with 4 of the 11 cases involving anaphylactic reactions (Figure 1). The TGA is aware of an additional 193 international reports which detail similar allergic-type reactions in relation to medicines containing Andrographis paniculata.
The New Zealand Medicines and Medical Devices Safety Authority has also published an alert communication in 2017[1] regarding 4 reports of allergic reaction in consumers taking products containing Andrographis paniculata. In the same year, the Royal Australasian College of Physicians published an abstract[2] highlighting that clinicians should be aware of the potential for Andrographis paniculata as a causative agent in anaphylaxis or other allergic reactions following ingestion of natural health products.
The TGA has continued to monitor medicines containing Andrographis paniculata and has received at least 14 adverse event reports per year associated with anaphylactic/allergic reactions which has further reinforced the concerns identified in the 2015 safety review. Although reporting of adverse events is compulsory for sponsors, adverse events are generally under-reported by consumers and health professionals, meaning the number of adverse events known to the TGA is likely to be below actual figures. As such, a warning statement is considered appropriate for listed medicines containing Andrographis paniculata to warn consumers of the risk of allergic reactions and anaphylaxis in some individuals.
What the TGA will do
The following label warning statement will be required on all listed medicines containing Andrographis paniculata:
'Andrographis may cause allergic reactions in some people. If you have a severe reaction (such as anaphylaxis) stop use and seek immediate medical attention' (or words to that effect).
The warning statement will be mandated through an update to the existing entry for Andrographis paniculata in the Therapeutic Goods (Permissible Ingredients) Determination. This Determination is expected to come into effect in December 2019 and will provide sponsors of existing listed medicines with a transition period until 2 May 2020 to comply with the new requirements. Any new listed medicines will need to comply immediately.
What sponsors of existing listed medicines need to do
Sponsors of existing listed medicines will need to ensure that medicines containing Andrographis paniculata that are released for supply from 2 May 2020 are compliant with the new requirements. Any new listed medicines need to comply with the new requirements immediately.
As this safety issue was raised in 2015, existing listed medicines may already have warning statements which reflect the new restrictions.
Sponsors of listed medicines affected by the variation to the Determination are encouraged to undertake corrective actions to address this issue immediately. Stock for existing listed medicines that has been released for supply prior to 2 May 2020 that does not have the new warning statement may continue to be sold. However, if an individual product is identified as having an unacceptable safety profile, further action may be taken.
It remains the sponsor's responsibility to ensure that their medicines remain safe for their intended use. If it appears that the safety of a medicine is unacceptable, the Secretary can cancel the medicine from the Register (see paragraph 30(2) of the Therapeutic Goods Act 1989).
Footnotes
| [1] | New Zealand Medicines and Medical Devices Safety Authority. (2017, 24/03/2017). Andrographis paniculata - potential risk for allergic reactions. Early Warning System - Alert Communication. Retrieved from https://medsafe.govt.nz/safety/EWS/2017/AndrographisPaniculata.asp# |
|---|---|
| [2] | Ahn, Y., & Brewerton, M. (2017). P2: When The Common Cold Turns Deadly: Anapylaxis With Andrographis paniculata Internal Medicine Journal, 47(S5), 5-5. doi:10.1111/imj.2_13578 |
| [2] | Hazell L, Shakir SAW. Under-reporting of adverse drug reactions: A systematic review Drug Saf 2006:29(5);385-396. |


