A face mask qualifies as a medical device when it is:
- used for the prevention of the transmission of diseases between people, or
- suitable for therapeutic use (surgical, clinical, medical use)
Unless exempt, all medical devices supplied in Australia must be included in the ARTG.
Non-sterile face masks that don’t make claims that qualify them as a medical device, are not regulated by the TGA.
These masks must not be advertised or labelled in a way that indicates:
- the mask prevents the spread of diseases between people, or
- for the use in healthcare (including aged care) settings.
Advertising
Therapeutic good advertising requirements apply to medical devices.
References to serious conditions, diseases are restricted and need TGA approval before use. Using words like "prevent, cure, treat, or diagnose serious conditions" is prohibited.
Advertisers can't say the goods are approved by the government. This is illegal.
See Restricted and prohibited representations to learn more.
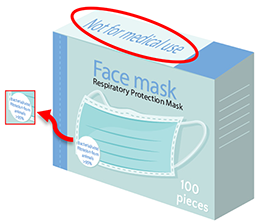
'Not for medical use'
In some cases, an individual or organisation may have purchased face masks for therapeutic use (e.g., in a clinical setting) based on their inclusion in the ARTG and subsequently received face masks with loose leaf information stating 'not for medical use'.
In these cases, you should consider the evidence and information about the intended purpose provided by the manufacturer to determine if the mask may be used in a clinical setting.
Be aware that a mask does not need to be suitable for medical use for it to be included in the ARTG.
For example, in a nonclinical setting, the mask might prevent transmission of diseases from spreading.
Changes to Chinese export regulation
The TGA is aware of loose-leaf inclusions in Mandarin of:
- 'non-medical',
- 'not for medical use' or
- 'not for surgical use' or similar descriptions in boxes of face masks.
April 2020, China announced changes to regulations for medical supplies and PPE.
Medical products for export now have to be registered with:
- the National Medical Products Administration (NMPA) or
- included in the 'white list' of internationally certified suppliers.
TGA doesn't have information on the requirements for either list.
The Chinese government decided that PPE products manufactured in China, but not by a manufacturer on either whitelist or NMPA list, were required to include labelling. Loose-leaf strips in Chinese language are often inserted into the product.
A 'not for medical use' label on a face mask doesn't mean it's not a 'medical device'.
This type of information does not indicate the mask does not meet the TGA's required quality and performance standards.
Interpreting labelling claims
The manufacturer's intended purpose is a key consideration when deciding if a face mask is a medical device. A face mask may still be a medical device even if it is labelled as 'not for medical use' as the manufacturer may intend it to be used for the prevention of the transmission of disease between people but in a non-surgical setting.
Below are examples of labelling and claims, manufacturer information, and how to interpret them.
| Claim/labelling | TGA interpretation | ARTG required? | Example |
|---|---|---|---|
'Not a medical device' No indication of preventing disease transmission. | The manufacturer does not intend the product to be used as a medical device. The product is not a medical device. | No |
|
There is a slip in the box of masks stating, 'not for medical use'. This slip may be a customs slip. The manufacturer intends the mask to prevent transmission of diseases between people. | The information from the manufacturer with respect to the intended purpose confirms the mask is a medical device. | Yes |
|
There is a slip/certificate in the box of masks stating, 'not for medical use'. This slip may be a customs slip. Manufacturer says intended purpose is general consumer use. | The information from the manufacturer with respect to the intended purpose confirms this is not a medical device. | No |
|
‘Not for medical use'. The manufacturer intends the mask to prevent transmission of diseases between people. | The information from the manufacturer with respect to the intended purpose confirms the mask is a medical device. It is not indicated for use in a clinical setting.as evidenced by the manufacturer's labelling. | Yes |
|
'Not for medical use'. Manufacturer says intended purpose is general consumer use. | This product is not intended to be used to prevent transmission of disease between persons, nor to be used in a clinical setting as evidenced by the manufacturer's labelling. | No |
|
'Not for medical use' and has indications of particle filtration. The manufacturer intends the mask to prevent transmission of diseases between people. | This product is not indicated for use in a clinical setting as evidenced by the manufacturer's labelling. The information from the manufacturer with respect to the intended purpose confirms the mask is a medical device. | Yes |
|
'Not for medical use' and has indications of particle filtration. The manufacturer has provided further information that the intended purpose is for use in an industrial setting. | This product is not indicated for use in a clinical setting as evidenced by the manufacturer's labelling. This product is indicated to prevent dust particle inhalation. | No |
|
'Not for medical use' Has indications of bacterial/viral particle filtration. The manufacturer intends the mask to prevent transmission of diseases between people. | The information from the manufacturer with respect to the intended purpose confirms that the mask is a medical device. However, it is not indicated for use in a clinical setting as evidenced by the manufacturer’s labelling. | Yes |
|
‘Not for medical use’ Has indications of bacterial/viral particle filtration. The manufacturer intends the mask to prevent microbial transmission from dirt to a person. | This product is not intended to prevent transmission of disease between persons. It is not a medical device. | No |
|
| ‘Not for medical use’ Has indications of bacterial/viral particle filtration to prevent transmission of disease from animal origin to human. | The manufacturer does not intend the mask to be used in clinical settings. As the prevention of transmission of disease is not for person to person it is not required to be included in the ARTG. | No |
|
| Labelled as a ‘surgical’ or ‘medical’ mask or respirator. | Suitable for use in surgery, or clinical, medical, or other health services. | Yes |
|
Labelled as a KN95 / N95 / P2. Intended to prevent transmission of disease between persons or to be used in a clinical setting. | Intended to prevent the transmission of disease between people and/or be suitable for use in surgery, clinical, medical, or other health services. | Yes |
|
Labelled as a KN95/N95/P2. The intended purpose is for use in an industrial setting. | This product is not intended to prevent the transmission of disease between people nor is it suitable for use in a clinical setting. | No |
|
Post-market review
We undertook a post-market review of face masks. The review validated the performance of face masks included in the ARTG, to ensure the safety of face mask users.
We will cancel any face masks found to be:
- not a medical device or
- non-compliant with the safety, quality, and performance requirements
Our list of cancelled ARTG entries for face masks has the latest cancellations.
We will not test products no longer included in the ARTG. We can only request samples of medical devices currently in the ARTG.
Cancelled from the ARTG
The ARTG entry for a face mask may be cancelled by:
- the sponsor (the person or company legally responsible for supplying the device in Australia) at any time, or
- the TGA if the product is not a medical device or if there is insufficient evidence to demonstrate the safety, performance, or quality of the device. In this case, the TGA will determine if a product defect notice is appropriate to ensure consumers are aware of the cancellation through the procedure for recalls, product alerts and product corrections (PRAC).
If you have a face mask initially supplied under an ARTG entry which has now been cancelled by the TGA or by the sponsor, you may continue to use it at your discretion.
Masks cancelled from the ARTG can't advertise with claims such as:
- for use in surgery, or clinical, medical or other health services;
- offering antimicrobial or anti-viral protection; or
- infection prevention or reduction between people, including against SARS-CoV-2 (COVID-19).
State and territory government officials
When jurisdictions buy face masks before cancellation, their distribution is not prohibited.
Whether those face masks are used if the entry is cancelled is at the discretion of the jurisdiction.
Reporting concerns
We have online forms for reporting any concerns or issues:
- Report a perceived breach or questionable practices, including the sale of unauthorised products.
- Report potentially false or misleading advertising.
- Report a problem with a medical device.
For frequently asked questions and more information about reporting medical device adverse events, see Medical device incident reporting and investigation scheme (IRIS).