Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
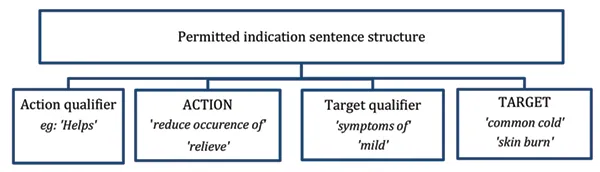
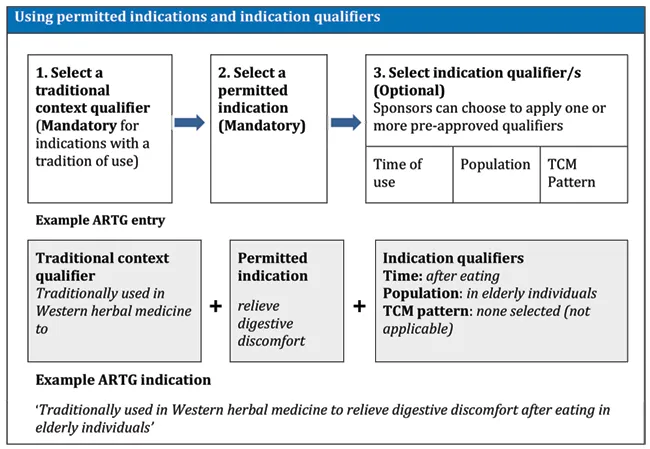
As listed medicines are not evaluated, the Therapeutic Goods Administration (TGA) uses a variety of mechanisms to help ensure that they are of high quality and used safely by consumers. One of these mechanisms is that listed medicines must only use indications drawn exclusively from a list of pre-approved 'permitted indications' and must comply with any requirements for the use of these indications in listed medicines.
This guidance provides information for sponsors on:
- what permitted indications are, including terminology and structure
- the use of permitted indications for listed medicines; and
- how to apply for inclusion of new indications on the permitted indications list.
For further information on the regulation of complementary medicines in Australia, please refer to the Australian Regulatory Guidelines for Listed Medicines and Registered Complementary Medicines.