Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Purpose
The Therapeutic Goods Act 1989 (the Act) and Therapeutic Goods (Medical Devices) Regulations 2002 (the Regulations) specify that:
- applications to include certain IVD medical devices (IVDs) in the ARTG must be selected for an application audit - an application audit assessment fee will be charged
- the TGA may also select any other application for inclusion for an application audit - an audit assessment fee will not be charged for these audits.
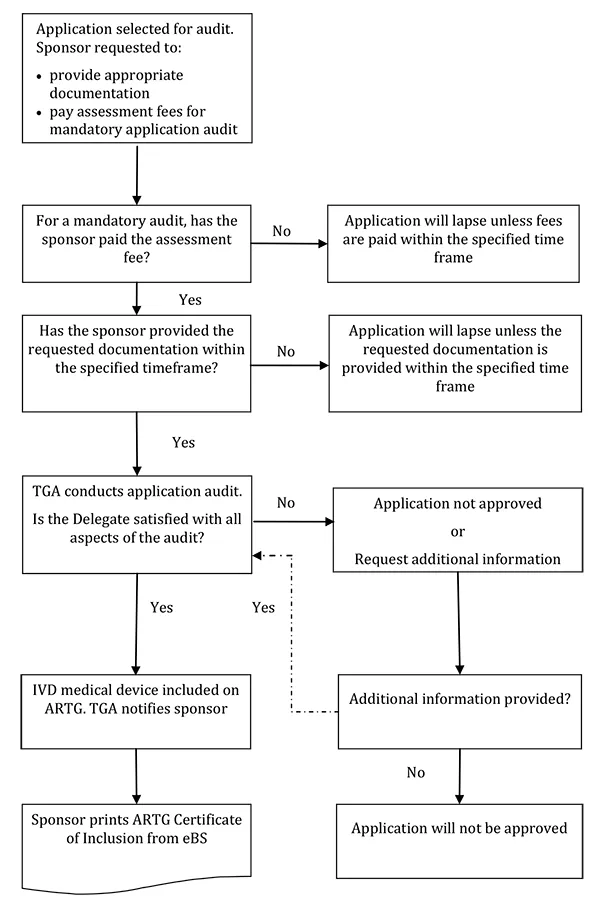
For an IVD, an application audit is primarily a review of the manufacturer's technical documentation and will often be referred to as a technical file review (TFR)1.
When conducting an application audit, the TGA may ask for any information or documents which may be relevant to demonstrating compliance with the Essential Principles for safety and performance, the conformity assessment procedure which has been applied by the manufacturer or information about advertising or supply of the IVD.
If an application audit is to be conducted, we will write to the sponsor who submitted the application to include the IVD on the ARTG advising:
- that the application has been selected to undergo an application audit;
- the documentation that the TGA requires the sponsor to provide;
- if applicable, the fee that is payable. The TGA will send a separate invoice formally requesting the payment - the invoice will provide the payment options and the due date for payment.
It should be noted that sponsors cannot submit the documentation before we formally writing to request it.