Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
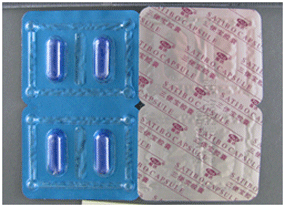
The Therapeutic Goods Administration (TGA) is aware that some Australian consumers have purchased the product SATIBO capsules over the internet, and that the product claims to be an "All natural Chinese herbal formula".
However, analysis untaken by the TGA laboratories in Australia has confirmed the presence of therapeutic quantities of tadalafil in these capsules. Tadalafil is the active ingredient in the Eli Lilly Australia Pty Ltd product Cialis, which is indicated for the treatment of erectile dysfunction in adult males. Also present in therapeutic quantities was hydroxyhomosildenafil, a derivative of sildenafil, the active ingredient in the Pfizer Australia Pty Ltd product Viagra, which is also indicated for the treatment of erectile dysfunction in adult males.
Tadalafil and hydroxyhomosildenafil are prescription-only substances in Australia, and may be harmful if taken without the supervision of a medical professional, particularly in patients with a history of cardiovascular disease.
SATIBO capsules have not been assessed by the TGA for quality, safety or efficacy as required under Australian legislation, and the place of manufacture is not approved by the TGA. In Australia, commercial supply of SATIBO capsules is illegal.
If you have purchased Satibo you are advised to cease using them and discard any remaining product. If you have concerns arising from your use of this product you should consult your health care practitioner.
Consumers are again advised to exercise extreme caution about purchasing medicines from unknown overseas Internet sites. Products purchased in this way may not meet the same standards of quality, safety and efficacy as those approved by the TGA for supply in Australia, and may contain undisclosed and potentially harmful ingredients.