Website maintenance took place on Thursday 30 April 2026. If you experience any issues, please contact us.
Consumers and health professionals are advised that LMT Surgical, in consultation with the TGA, is issuing a hazard alert for all sizes and lots of its PyroTitan humeral resurfacing arthroplasty device.
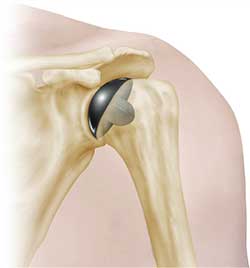
PyroTitan humeral resurfacing arthroplasty devices are used in humeral resurfacing procedures.
It has been identified that there is potential for PyroTitan humeral resurfacing arthroplasty devices to break after being implanted. If this occurs, revision surgery will be necessary.
In most confirmed cases, breakage has occurred within a year of the device being implanted and has been the result of excessive loads being put on the device.
Excessive loads may occur during high impact activities (such as hammering; free weights; and contact sports, such as football) or as a result of sudden trauma.
Information for consumers
LMT Surgical has written to surgeons who have implanted PyroTitan humeral resurfacing arthroplasty devices, providing further information about the above issue and advice on how to treat affected patients.
If you have had a shoulder replacement and experience unexpected pain in your shoulder or any other unusual symptoms, or if you have any questions or concerns about the above issue, you should contact your surgeon or the hospital where the surgery was performed.
Information for health professionals
Patients who have had a shoulder replacement and who have any questions or concerns about the above issue should be referred to their surgeon or the hospital where the surgery was performed.
Information for orthopaedic surgeons
LMT Surgical has written to surgeons who have implanted PyroTitan humeral resurfacing arthroplasty devices, providing further information about the above issue and advice on how to treat affected patients.
LMT Surgical advises that excessive loads placed on the device through high impact activities or sudden trauma can damage the artificial joint, particularly in the presence of poor bone stock.
High impact activity may cause loosening or fracture of the device.
A breakage can result in glenohumeral joint pain and possible damage to the surrounding tissue.
This hazard alert is being undertaken because 3% of implanted devices worldwide have experienced breakage.
Two of the devices returned to the manufacturer for examination have been found to have sub-surface fractures, which is believed to have contributed to the high rate of breakage.
Future supply of PyroTitan humeral resurfacing arthroplasty devices in Australia will not occur before appropriate corrective actions have been implemented.
It is expected that the corrective action(s) will take several months to complete.
Additional information
The PyroTitan humeral resurfacing arthroplasty devices affected by this hazard alert are:
| Product code | Description |
|---|---|
| CHRA-910-38/14-WW | PyroTitan Size 38-14 |
| CHRA-910-41/15-WW | PyroTitan Size 41-15 |
| CHRA-910-41/18-WW | PyroTitan Size 41-18 |
| CHRA-910-44/16-WW | PyroTitan Size 44-16 |
| CHRA-910-44/19-WW | PyroTitan Size 44-19 |
| CHRA-910-47/17-WW | PyroTitan Size 47-17 |
| CHRA-910-47/20-WW | PyroTitan Size 47-20 |
| CHRA-910-50/18-WW | PyroTitan Size 50-18 |
| CHRA-910-50/21-WW | PyroTitan Size 50-21 |
| CHRA-910-53/19-WW | PyroTitan Size 53-19 |
| CHRA-910-53/22-WW | PyroTitan Size 53-22 |
| CHRA-910-56/21-WW | PyroTitan Size 56-21 |
Reporting problems
Consumers and health professionals are encouraged to report problems with medical devices. Your report will contribute to the TGA's monitoring of these products. For more information see the TGA Incident Reporting and Investigation Scheme (IRIS).
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medical device.

