Consumers and health professionals are advised that Medtronic, in consultation with the TGA, is issuing a hazard alert for its Medtronic RestoreSensor implantable neurostimulators.
A neurostimulation device
used for spinal cord
and peripheral nerve
stimulation therapies
RestoreSensor implantable neurostimulators can manipulate nerve pathways to reduce chronic pain. The implant is placed under a patient's skin to deliver electrical impulses to the spinal cord, which act to block pain signals from going to the brain.
Medtronic has confirmed four instances of loss of therapeutic effect worldwide during recharging of RestoreSensor implantable neurostimulator devices, equal to a rate of 0.007%. To date, there have been no reports of this issue occurring in Australia.
Potentially affected model numbers are:
- 37714 (ARTG 177594)
- 97714 (ARTG 215750)
By design, stimulation therapy turns off when the internal battery's voltage depletes below a certain level. Medtronic found that a key factor in the reported incidents was insufficient coupling ('Charging Efficiency') between the external recharger and the implanted neurostimulator during recharge sessions. This led to inadequate recharge of the battery and loss of stimulation therapy in affected devices. Should patients experience a loss of stimulation their underlying symptoms will return. Patients will need to visit their treating health professional to have therapy restored.
Medtronic is assessing the best approach to permanently resolve the issue as quickly and safely as possible and in the meantime has provided instructions on what to do to avoid encountering this issue (for more detail see 'Information for consumers' below).
Information for consumers
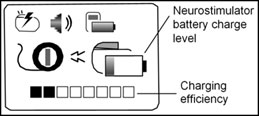
Where to find the Charging Efficiency
and Battery Charge Level indicators
on the display
Patients are advised to follow the current recharge instructions, paying particular attention to 'Charging Efficiency' and 'Battery Charge Level' indicators on the recharger:
- Check the neurostimulator battery charge level at least once a day, or more frequently as needed.
- Keep the neurostimulator sufficiently charged to maintain therapy. It can be charged at any time; you do not need to wait for a low battery message.
- During neurostimulator recharging, monitor the 'Charging Efficiency' indicator and adjust the antenna to obtain as many solid black boxes as possible. If six or more boxes are empty, adjust the antenna to improve the signal strength between the neurostimulator and recharger.
- During recharging, ensure the neurostimulator's 'Battery Charge Level' reaches at least 25% before ending the charge session. However, a full battery charge is preferred.If charging efficiency is allowed to stay too low and the battery is not recharged enough to restart stimulation therapy the battery can enter an 'over-discharge' state. If this occurs patients will experience a loss of stimulation and their underlying symptoms will return. Patients will need to visit their treating health professional to have therapy restored.
If a device enters an 'over-discharge' state three times it will stop functioning.
If you or a patient you care for experiences difficulty in recharging a neurostimulator, or an unexpected loss of therapy for any reason, you should contact your managing health professional immediately.
Medtronic Australasia has written to health professionals who have implanted spinal cord stimulation models affected by the hazard alert, or are managing patients who have these devices, providing further information.
Information for health professionals
If you are treating a patient who has a spinal cord stimulation device implanted, reassure them that problems associated with this issue are rare.
If a patient has any questions or concerns about this issue, refer them to their managing surgeon or neurologist.
Information for surgeons and pain specialists
Medtronic Australasia has written to surgeons and pain specialists who have implanted spinal cord stimulation models affected by the hazard alert, or are managing patients who have these devices, providing further information.
At their next scheduled visit, advise patients of this issue and reassure them that problems associated with it are rare. Advise patients to follow the current recharge instructions, paying particular attention to 'Charging Efficiency' and 'Battery Charge Level' indicators on the recharger, and notify them of the instructions listed in the 'Information for consumers' section above.
If an insufficient recharge coupling is continued and the battery is not recharged enough to restart stimulation therapy, the battery can discharge to an 'over-discharge' state. Under normal usage conditions, the patient has 30 days to recharge the device to avoid experiencing a battery 'over-discharge' stoppage. If insufficient recharge coupling is continued, the time period is reduced to 1-2 days.
Once a device is in an 'over-discharge' state, therapy is interrupted and can only be restored using the 'Physician Recharge Mode' of the recharger. This interruption could lead to a return of patient symptoms. The device will also require more frequent recharging as a result of reduced battery capacity and performance after an 'over-discharge' event.
There is also the potential for patients to experience overstimulation or stimulation in the wrong area following an 'over-discharge' recovery that causes pain, discomfort, and/or shaking. As described in labelling, if three occurrences of 'over-discharge' occur, the neurostimulator will cease functioning, and must be replaced to restore therapy.
If you have any questions or concerns regarding this issue, contact Medtronic Australasia customer service on 1800 668 670.
Reporting problems
Consumers and health professionals are encouraged to report problems with medical devices. Your report will contribute to the TGA's monitoring of these products. For more information see the TGA Incident Reporting and Investigation Scheme (IRIS).
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medical device.

