 Consumers and health professionals are advised that Medtronic Australasia, in consultation with the TGA, is undertaking a recall for product correction of Medtronic model 37751 rechargers due to an error that causes the recharger to become unresponsive.
Consumers and health professionals are advised that Medtronic Australasia, in consultation with the TGA, is undertaking a recall for product correction of Medtronic model 37751 rechargers due to an error that causes the recharger to become unresponsive.
The device may be used with or without a power supply being connected. This error only occurs when it is used to recharge without a power supply being connected.
To address this issue, Medtronic Australasia is providing information to health professionals, including instructions for patients on how to prevent this problem and restore functionality to unresponsive rechargers.
Medtronic model 37751 rechargers are sold in kits for certain neurostimulation systems, including deep brain stimulation (DBS) and spinal cord stimulation (SCS) devices.
Neurostimulation devices are implantable, programmable medical devices that deliver electrical stimulation to specific parts of the patient's brain, spinal cord or peripheral nervous system to help treat various conditions, including chronic pain, movement disorders, epilepsy and Parkinson's disease.
Medtronic model 37751 rechargers are used by DBS and SCS patients who have the following devices implanted:
- DBS - Medtronic Activa RC (Model 37612)
- SCS - Restore (Model 37711)
- SCS - RestoreUltra (Model 37712)
- SCS - RestoreAdvanced (Model 37713)
- SCS - RestoreSensor (Model 37714)
- SCS - RestoreUltra SureScan MRI (Model 97712)
- SCS - RestoreAdvanved SureScan MRI (Model 97713)
- SCS - RestoreSensor SureScan MRI (Model 97714).
It has been identified that Medtronic model 37751 rechargers may stop functioning and this loss of function is signalled by a blank display screen combined with a beep every five seconds. This occurs only when the recharger is used while disconnected from a power supply.
If this occurs, the recharger must be reset as it will no longer recharge the neurostimulator battery. Once the neurostimulator battery becomes fully depleted, the patient will experience a loss of therapy and possibly a return of disease specific symptoms. For a subset of patients receiving DBS therapy, in rare instances, this could lead to life threatening injury or death. However, as of 27 October 2016, there have been no reports of life threatening injury or death associated with this issue.
If the neurostimulator battery is allowed to remain fully depleted it may overdischarge, resulting in permanent reduction in battery capacity and the need to recharge more frequently.
Rechargers manufactured since November 2014 are more susceptible to this problem (approximately 2% of all rechargers manufactured and sold after November 2014, compared to 0.2% of rechargers manufactured before November 2014).
Information for consumers
If you or someone you provide care for has a neurostimulator that uses a Medtronic model 37751 recharger, be aware of this issue.
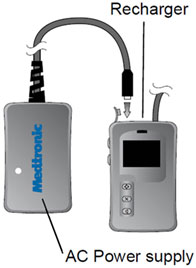
To prevent this error, the recharger should be plugged into the AC power supply (by aligning the white triangles) prior to starting a recharging session of your neurostimulator. The recharger should remain connected to the AC power supply until the recharging session has finished. Keeping the recharger plugged into the AC power supply during recharging will prevent the error state from occurring.
The AC power supply does not need to be plugged into a power outlet if the recharger is charged.
If the recharger is not connected to the AC power supply during a recharging session of the neurostimulator and the unresponsive error occurs (recharger stops functioning, has a blank display screen and beeps every five seconds), you can contact Medtronic Neuromodulation Patient Services at 1800 652 972 for instructions on how to reset your device.
This issue can recur after reset if the recharging instructions are not completely followed for future recharging sessions.
You should contact your surgeon or neurologist if you experience a return of symptoms.
If you have any questions or concerns about this issue, talk to your health professional.
Information for all health professionals
If you are treating a patient who has a rechargeable neurostimulator used for DBS or SCS therapy, be aware of this issue.
If your patient's recharger stops functioning and beeps every five seconds, or if they have any questions or concerns about this issue, refer them to their surgeon or neurologist.
Information for surgeons and neurologists
Medtronic Australasia has written to surgeons and neurologists who have implanted neurostimulators that use Medtronic model 37751 rechargers, or are managing patients who have these devices, providing further information including a patient notification document.
Until a permanent solution for this issue is in place, Medtronic Australasia recommends that you discuss this issue with any current or new patient implanted with these devices and provide them with a copy of the patient notification.
Ensure that they are aware of and understand the instructions outlined in the above 'Information for consumers' section on how to prevent this issue or reset the recharger if it occurs.
If you have any questions or concerns regarding this issue, contact Medtronic Australasia customer service on 1800 668 670.
Reporting problems
Consumers and health professionals are encouraged to report problems with medical devices. Your report will contribute to the TGA's monitoring of these products. For more information see the TGA Incident Reporting and Investigation Scheme (IRIS).
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medical device.

