Consumers and health professionals are advised that TGA has received reports of patients being infected by contaminated Meditech ultrasound gel being used in intensive care and radiology units. The sponsor is undertaking a recall of all lots of the gel (recall number: RC-2017-RN-00631-1).
The affected gel is provided as part of a kit which includes a sterile ultrasound probe cover and has been used in some States and Territories for specialist catheter insertion. The presentation of this gel means that it is not used for common ultrasound procedures such as in obstetrics.
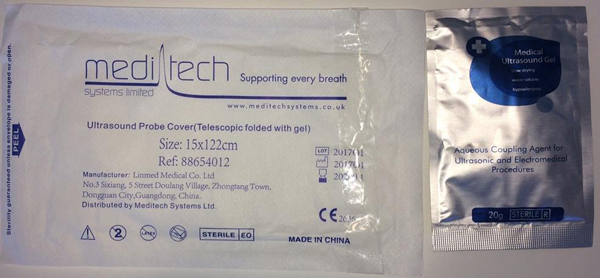
The ultrasound gel sachets pictured below may also be supplied in other kits or on their own. Health professionals and other users of this gel should cease use of this gel and source another product.
Burkholderia cenocepacia bacteria have been isolated from the blood cultures of seven patients from intensive care units in Queensland and two patients in the ACT since the end of March 2017. This organism is commonly found in the environment, but is not usually found in blood cultures.
All of these patients had recently had central lines placed under ultrasound guidance using an ultrasound probe cover and gel manufactured by Linmed, a Chinese company.
All patients are recovering from the infection, although many have other serious health issues.
Testing by the microbiology laboratories at affected hospitals found that several samples of the gel in the Meditech kits, which should be sterile, were heavily contaminated with Burkholderia cenocepacia.
In addition, Burkholderia cenocepacia has been isolated from fluid in the abdominal cavity of a patient at another Queensland hospital where the same Meditech covers and gel are used.
Samples of the ultrasound gel will be tested by the TGA in its own laboratories.
Information for consumers
The affected gels are only used in some health facilities undertaking certain procedures.
All State and Territory Health departments have been notified of this issue and are advising their hospitals to cease using the affected ultrasound gel. They are monitoring/treating patients who may have been exposed.
Burkholderia cenocepacia infections are treatable but pose additional risks in people with cystic fibrosis where such infections can be serious and even life-threatening.
If you or someone you provide care for is hospitalised and may have undergone a procedure using affected gel, be aware that the health professionals who work in these areas are highly trained and have protocols to deal with the potential issues that have been outlined.
If you have any questions or concerns about this issue, speak to the hospital staff.
Information for health professionals
Approximately 2500 ultrasound probe cover kits have been supplied by Meditech that were manufactured by Linmed, to 13 hospitals and distributors in QLD, ACT, NSW and WA.
Health facilities are advised to immediately cease using Meditech ultrasound gel products and to quarantine unused stock and when notified of the recall follow the instructions regarding the disposal or return of the gel.
Facilities should be alert to patients who may have been exposed and follow advice from their infection control teams and state or territory health departments.
Further advice regarding this issue and associated recall will be provided as more information becomes available.
Reporting problems
Consumers and health professionals are encouraged to report problems with medical devices. Your report will contribute to the TGA's monitoring of these products. For more information see the TGA Incident Reporting and Investigation Scheme (IRIS).
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medical device.

