Consumers and health professionals are advised that Medtronic Australasia, in consultation with the TGA, has issued a hazard alert regarding InterStim and InterStim II neurostimulation devices, which are used for sacral nerve stimulation therapies.
Medtronic Australasia is also undertaking a recall for product correction to update the instructions for use.
InterStim and InterStim II neurostimulation devices are implantable, programmable medical devices that deliver electrical stimulation to sacral nerves to help treat various conditions, including overactive bladder, urinary retention and faecal incontinence.
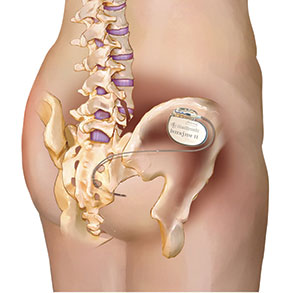
It has been identified that enabling the cycling feature in InterStim (model 3023) and InterStim II (model 3058) neurostimulation devices may lead to premature battery depletion.
If premature battery depletion occurs, the patient may have to undergo surgery earlier than would normally be expected to address the issue.
Medtronic Australasia is updating the labelling for affected devices and is providing updated information to assist health professionals manage patients who are receiving sacral nerve stimulation using these devices.
As of 27 January 2015, there have been no reports in Australia of patients experiencing problems as a result of this issue.
Information for consumers
Medtronic Australasia has written to health professionals who have implanted InterStim and InterStim II neurostimulation devices, or are managing patients who have these devices, providing further information.
If you have an InterStim or InterStim II neurostimulation device, but do not use the cycling feature, you are not affected by this issue.
If your InterStim or InterStim II neurostimulation device uses the cycling feature, continue to check your battery status as normal. Instructions on how to check battery status can be found in the patient programmer or recharger system manual.
If you have any questions or concerns regarding this issue, contact your managing health professional.
Information for all health professionals
If you are treating a patient who has an InterStim or InterStim II neurostimulation device and they have any questions or concerns about this issue, refer them to their managing surgeon or neurologist.
Information for surgeons and neurologists
If you are managing a patient who has an InterStim or InterStim II neurostimulation device, at their next scheduled visit, advise them of this issue and remind them to continue checking their battery status as normal. Instructions on how to check battery status can be found in the patient programmer or recharger system manual.
References to the use of cycling for improved battery longevity, when compared to continuous mode, are not accurate for every combination of programmable parameters.
If cycling is being used solely to improve device battery longevity, the information below should be considered.
For patients with cycling enabled and pulse amplitudes greater than or equal to 1 Volt, the following cycling intervals may improve device battery longevity when compared to programming in continuous mode:
- cycling intervals greater than or equal to two seconds ON and greater than or equal to two seconds OFF, without SoftStart/Stop enabled; or
- cycling intervals greater than or equal to 60 seconds ON and greater than or equal to 60 seconds OFF, with SoftStart/Stop enabled and programmed at greater than four seconds.
Cycling at intervals less than those noted above with pulse amplitudes less than or equal to 1 Volt, may result in reduced device battery longevity, when compared to continuous mode. At any time while cycling is enabled, the longevity estimate provided by labelling and the N’Vision Clinician Programmer may not be accurate.
If you have any questions or concerns regarding this issue, contact Medtronic Australasia customer service on 1800 668 670.
Reporting problems
Consumers and health professionals are encouraged to report problems with medical devices. Your report will contribute to the TGA’s monitoring of these products. For more information see the TGA Incident Reporting and Investigation Scheme (IRIS).
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medical device.

