Consumers and health professionals are advised that HeartWare, in consultation with the TGA, has issued a hazard alert for its HeartWare Ventricular Assist Device (HVAD) due to potential issues with fluid or foreign material entering the driveline and triggering electrical faults.
HeartWare is also recalling affected devices that have not yet been implanted.
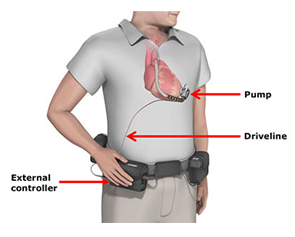
The HVAD system takes over some or all of the pumping function of the heart. The device includes a pump which is attached to the patient's heart and is connected via a driveline to an external controller that regulates the pump's function and monitors the system. The system is implanted either in patients awaiting heart transplants or as an alternative to transplantation.
Potentially affected devices carry model number 1104 with serial numbers lower than HW25838 (all affected serial numbers start with the prefix HW).
HeartWare has advised that this issue has been reported in approximately 5% of implanted devices and it most commonly occurs within the first 30 days of implantation. There were two reported cases of this issue occurring in Australia in the two years to 30 August, 2016. In both cases qualified HeartWare personnel undertook successful driveline cleaning and controller exchange procedures in controlled clinical settings. The patients were discharged home in stable conditions and subsequently there have been no reported electrical faults for these patients.
Information for consumers
If you or someone you care for has an implanted HeartWare device check the model and serial numbers. If you have an affected model and the serial number is lower than HW25838 you should contact your cardiologist or the hospital where the operation was performed for advice. If you are unsure of the model or serial numbers on your device, contact your cardiologist.
A patient with an implanted device who experiences electrical faults due to driveline connector contamination should see a 'medium' alarm on their controller display. The controller will provide an audible alarm that increases in volume and a flashing yellow alarm indicator. Additionally, the controller display will provide the text message 'Electrical Fault - Call'. If this fault is displayed you should contact your cardiologist promptly.
Your cardiologist can arrange for a qualified HeartWare technician to undertake remedial action on the device.
If you have any questions or concerns regarding this issue, contact your cardiologist.
Information for all health professionals
If you are treating a patient who has a HeartWare implant, advise them of this issue. Assist the patient to identify if they have an affected device (model number 1104 with serial numbers lower than HW25838).
If your patient does not have an affected device, reassure them that they are not affected by this recall notice.
If the patient does have a device affected by this recall, refer them to the cardiologist and/or hospital where the procedure was performed for further advice.
If they have any further questions or concerns about the above issue, refer them to their cardiologist.
Information for cardiologists and hospitals
HeartWare has written to cardiologists who have implanted the HeartWare HVAD system and to HeartWare clinicians, advising them of this issue and the recall procedure.
HeartWare does not advise removal of affected devices which have already been implanted into a patient. Patients with implanted devices experiencing electrical faults due to driveline connector contamination should see a 'medium' alert on their controller display. The issue can be addressed by a qualified HeartWare technician undertaking a connector cleaning procedure.
If you have further questions or concerns regarding these issues, contact HeartWare on (02) 8078 6164 or email: csaustralia@heartware.com.
Reporting problems
Consumers and health professionals are encouraged to report problems with medical devices. Your report will contribute to the TGA's monitoring of these products. For more information see the TGA Incident Reporting and Investigation Scheme (IRIS).
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medical device.

