Consumers and health professionals are advised that Wright Medical Australia, in consultation with the TGA, has issued a hazard alert regarding specific lots of its Claw and Claw II plates. Wright Medical Australia is also recalling unused stocks of the affected lots.
Claw and Claw II plates comprise plates and locking screws of various lengths, which are used in surgery to reposition and stabilise bones in the foot.
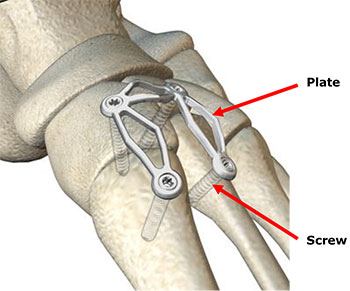
It has been identified that the stainless steel used in Claw and Claw II plates from the affected lots did not meet the manufacturing requirements, resulting in reduced strength and increased risk of warping under stress. If this occurs, the device could fail.
If this problem occurred during surgery, it would be corrected immediately. However, surgery time would be increased.
If an affected device has been implanted and did not fail during surgery, there is a remote possibility that it could fail before fusion of the bones occurs. If this happens, revision surgery will be required.
As of 25 August 2014, there have been no reports of patient injury or the need for revision surgery as a result of this issue.
Information for consumers
Wright Medical Australia has contacted surgeons to provide further information about this issue, including advice on how to treat affected patients.
If you have had a Claw or Claw II plate implanted in your foot, the likelihood of you experiencing any problems relating to this issue after your operation is very low. If the problem occurred during implantation, it would have been corrected immediately.
You are reminded to follow all post-operative instructions, including to avoid placing weight on the foot until advised by your surgeon that it has adequately healed.
If you have any questions or concerns about this issue, contact your surgeon.
Information for all health professionals
If you are treating a patient who has had fusion surgery on a foot and is concerned about this matter, reassure them that the likelihood of them experiencing any problems relating to this issue after their operation is very low. Advise them to follow all post-operative instructions.
If they continue to have questions or concerns, refer them to their surgeon.
Information for surgeons
Wright Medical Australia has contacted surgeons to provide further information about this issue, including advice on how to treat affected patients and details of the recall process.
Check any unused Claw and Claw II plates you have in stock to see if they are from an affected lot. If they are, those plates should not be used. Instead, they should be quarantined before being returned to Wright Medical Australia.
The above issue may have been observed during surgery as reduced torque resistance or increased bending/compression.
If you are treating a patient who has had a Claw or Claw II plate implanted, reassure them that the likelihood of them experiencing any problems relating to this issue after their operation is very low.
Advise patients to follow the post-operative instructions, including to not bear weight on the foot before fusion occurs.
Prophylactic revision surgery is not recommended.
Affected lots
The affected lot and item numbers are:
| Lot number | Item number | Description |
|---|---|---|
| 1509955 | 40130130 | Claw 3.5mm Plate 2 Hole 30mm |
| 1487673 | 40240215 | Claw II 2 Hole 15mm |
| 1487674 | 40240215 | Claw II 2 Hole 15mm |
| 1487675 | 40240215 | Claw II 2 Hole 15mm |
| 1487676 | 40240215 | Claw II 2 Hole 15mm |
| 1490003 | 40240215 | Claw II 2 Hole 15mm |
| 1512150 | 40240215 | Claw II 2 Hole 15mm |
| 1512151 | 40240215 | Claw II 2 Hole 15mm |
| 1519497 | 40240215 | Claw II 2 Hole 15mm |
| 1519498 | 40240215 | Claw II 2 Hole 15mm |
| 1522019 | 40240215 | Claw II 2 Hole 15mm |
| 1522020 | 40240215 | Claw II 2 Hole 15mm |
| 1523747 | 40240215 | Claw II 2 Hole 15mm |
| 1523748 | 40240215 | Claw II 2 Hole 15mm |
| 1537072 | 40240215 | Claw II 2 Hole 15mm |
| 1537073 | 40240215 | Claw II 2 Hole 15mm |
| 1537162 | 40240215 | Claw II 2 Hole 15mm |
| 1539399 | 40240215 | Claw II 2 Hole 15mm |
| 1541615 | 40240215 | Claw II 2 Hole 15mm |
| 1541616 | 40240215 | Claw II 2 Hole 15mm |
| 1543148 | 40240215 | Claw II 2 Hole 15mm |
| 1543149 | 40240215 | Claw II 2 Hole 15mm |
| 1490004 | 40240220 | Claw II 2 Hole 20mm |
| 1490005 | 40240220 | Claw II 2 Hole 20mm |
| 1490199 | 40240220 | Claw II 2 Hole 20mm |
| 1490200 | 40240220 | Claw II 2 Hole 20mm |
| 1490201 | 40240220 | Claw II 2 Hole 20mm |
| 1490202 | 40240220 | Claw II 2 Hole 20mm |
| 1518483 | 40240220 | Claw II 2 Hole 20mm |
| 1519493 | 40240220 | Claw II 2 Hole 20mm |
| 1519494 | 40240220 | Claw II 2 Hole 20mm |
| 1519495 | 40240220 | Claw II 2 Hole 20mm |
| 1522021 | 40240220 | Claw II 2 Hole 20mm |
| 1522898 | 40240220 | Claw II 2 Hole 20mm |
| 1522899 | 40240220 | Claw II 2 Hole 20mm |
| 1522900 | 40240220 | Claw II 2 Hole 20mm |
| 1523735 | 40240220 | Claw II 2 Hole 20mm |
| 1523736 | 40240220 | Claw II 2 Hole 20mm |
| 1526519 | 40240220 | Claw II 2 Hole 20mm |
| 1526520 | 40240220 | Claw II 2 Hole 20mm |
| 1526521 | 40240220 | Claw II 2 Hole 20mm |
| 1538760 | 40240220 | Claw II 2 Hole 20mm |
| 1538761 | 40240220 | Claw II 2 Hole 20mm |
| 1539402 | 40240220 | Claw II 2 Hole 20mm |
| 1540715 | 40240220 | Claw II 2 Hole 20mm |
| 1540716 | 40240220 | Claw II 2 Hole 20mm |
| 1540717 | 40240220 | Claw II 2 Hole 20mm |
| 1542703 | 40240220 | Claw II 2 Hole 20mm |
| 1542704 | 40240220 | Claw II 2 Hole 20mm |
| 1542776 | 40240220 | Claw II 2 Hole 20mm |
| 1543491 | 40240220 | Claw II 2 Hole 20mm |
| 1543891 | 40240220 | Claw II 2 Hole 20mm |
| 1490367 | 40240225 | Claw II 2 Hole 25mm |
| 1490368 | 40240225 | Claw II 2 Hole 25mm |
| 1512152 | 40240225 | Claw II 2 Hole 25mm |
| 1521232 | 40240225 | Claw II 2 Hole 25mm |
| 1523734 | 40240225 | Claw II 2 Hole 25mm |
| 1523746 | 40240225 | Claw II 2 Hole 25mm |
| 1537160 | 40240225 | Claw II 2 Hole 25mm |
| 1537161 | 40240225 | Claw II 2 Hole 25mm |
| 1539401 | 40240225 | Claw II 2 Hole 25mm |
| 1541613 | 40240225 | Claw II 2 Hole 25mm |
| 1541614 | 40240225 | Claw II 2 Hole 25mm |
| 1542652 | 40240225 | Claw II 2 Hole 25mm |
| 1542774 | 40240225 | Claw II 2 Hole 25mm |
| 1542775 | 40240225 | Claw II 2 Hole 25mm |
| 1487670 | 40240230 | Claw II 2 Hole 30mm |
| 1487671 | 40240230 | Claw II 2 Hole 30mm |
| 1487672 | 40240230 | Claw II 2 Hole 30mm |
| 1512149 | 40240230 | Claw II 2 Hole 30mm |
| 1519496 | 40240230 | Claw II 2 Hole 30mm |
| 1523745 | 40240230 | Claw II 2 Hole 30mm |
| 1534796 | 40240230 | Claw II 2 Hole 30mm |
| 1537159 | 40240230 | Claw II 2 Hole 30mm |
| 1539400 | 40240230 | Claw II 2 Hole 30mm |
| 1540714 | 40240230 | Claw II 2 Hole 30mm |
| 1512153 | 40242020 | Claw II Straight 20 x 20mm |
| 1518480 | 40242020 | Claw II Straight 20 x 20mm |
| 1532497 | 40242020 | Claw II Straight 20 x 20mm |
| 1538075 | 40242020 | Claw II Straight 20 x 20mm |
| 1538076 | 40242020 | Claw II Straight 20 x 20mm |
| 1538332 | 40242020 | Claw II Straight 20 x 20mm |
| 1513144 | 40242025 | Claw II Straight 20 x 25mm |
| 1518479 | 40242025 | Claw II Straight 20 x 25mm |
| 1534798 | 40242025 | Claw II Straight 20 x 25mm |
| 1538331 | 40242025 | Claw II Straight 20 x 25mm |
| 1543489 | 40242025 | Claw II Straight 20 x 25mm |
| 1517327 | 40242525 | Claw II Straight 25 x 25mm |
| 1538333 | 40242525 | Claw II Straight 25 x 25mm |
| 1543490 | 40242525 | Claw II Straight 25 x 25mm |
| 1543488 | 40S20020 | Claw Plate 20mm Interaxis |
Reporting problems
Consumers and health professionals are encouraged to report problems with medical devices. Your report will contribute to the TGA's monitoring of these products. For more information see the TGA Incident Reporting and Investigation Scheme (IRIS).
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medical device.

