Consumers and health professionals are advised that Fit-BioCeuticals, in consultation with the TGA, is recalling all current batches of Blackmores Professional Duo Celloids S.C.F. tablets due to the potential for contamination with mould.
Blackmores Professional Duo Celloids S.C.F. is a complementary medicine that people use as it may assist bone formation.
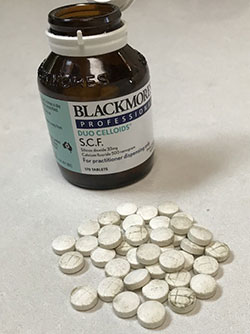
Mould is visible on these affected tablets.
Affected batch numbers are: 292798, 294442, 295592, 296051, 297013, 298225, 298013 and 296052.
The product has been identified as having a microbial mould growth on the tableted product which appears to be developing after some time in storage. The mould has been identified as a strain of Penicillium species. This is visible to the naked eye as black or brown patchy spots.
Penicillium mould can cause an adverse reaction, including an allergic reaction.
No other products are affected by this recall.
Information for consumers
If you or somebody you care for uses Blackmores Professional Duo Celloids S.C.F. tablets stop using it immediately.
You can return the unused product to the place of purchase for a refund.
For further information you can contact the customer service of Blackmores Professional's parent company, BioCeuticals, on 1300 650 455 or email cs@bioceuticals.com.au.
Information for health professionals
Blackmores Professional has written to health professionals regarding this issue.
If you are aware of a patient using this product advise them to stop immediately and return the product for a refund.
For pharmacists to initiate the return of any affected batches contact BioCeuticals Customer Service on 1300 650 455 for further details.
For further information you can contact the customer service of Blackmores Professional's parent company, BioCeuticals, on 1300 650 455 or email cs@bioceuticals.com.au.
Consumers and health professionals are encouraged to report problems with medicines or vaccines. Your report will contribute to the TGA's monitoring of these products.
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medicine or vaccine.

