Consumers and health professionals are advised that a recently published Australian study suggests a higher incidence of first hospitalisation due to heart failure among a group of male patients with ASR XL metal-on-metal (MoM) hip implants when compared with a similar group who had received a metal-on-polyethylene (MoP) implant.
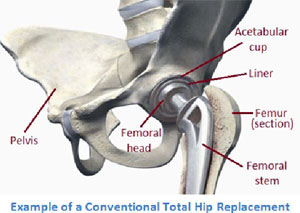
The ASR XL is a type of MoM hip implant in which the two moving surfaces in the joint are both made of metal, whereas in MoP implants one of the surfaces is made of a plastic polymer.
There is a growing body of evidence that because the two moving parts of a MoM hip implant wear against each other they can release trace amounts of cobalt and chromium (Co/Cr), especially during the first 18-months or so after the operation.
The latest study has several limitations, and the authors found no evidence of a higher rate of heart failure in either female patients with ASR XL or in patients of either gender who had another type of MoM implant compared with patients who received MoP implants.
For a full discussion of the study and its limitations see the 'Background' section below.
While the study reports that a higher proportion of male patients with ASR XL implants were hospitalised for heart failure after their hip replacements, there is nothing that reveals why this may be the case.
More investigation will be needed to establish whether having an ASR XL implant causes a higher incidence of heart failure.
Due to the limitations, the implications of the results of the study for the follow up of patients who have an ASR XL implant are highly uncertain.
The TGA is facilitating another study using information for a much larger group of patients. It is hoped that the results of the upcoming study will reduce the uncertainty.
The TGA and other regulatory agencies continue to actively look for further evidence of health issues associated with ASR XL and other MoM implants.
Information for consumers
If you or somebody you provide care currently has an ASR XL hip implant, note the following:
- A link between the ASR XL implant in elderly men and heart failure has been suggested but has NOT been confirmed.
- In women in particular, the study found that there is no connection between having an ASR XL hip replacement and heart disease.
- The TGA is actively investigating this issue and the potential implications of this study in an Australian cohort. Any developments in this area will be published on the TGA website.
- If you are concerned see your GP and/or orthopaedic surgeon and discuss what follow up may be required in your specific case.
If you have a MoM hip implant that is not an ASR XL implant, note the following:
- This new data does not confirm a connection between MoM hip implants and an increased risk of heart failure.
- Continue on your current medical follow up arrangements. There is no evidence to suggest that any change to follow up is required.
- If you are not sure about the type of hip implant that you have or if you are concerned about this issue, speak to your GP.
Information for health professionals
If you are treating a patient who has an ASR XL total hip replacement:
- Consider monitoring patients for early signs of heart failure through clinical examination and imaging including echocardiogram, especially if there are additional risk factors.
- For patients with current symptoms of hip pain that may suggest adverse local tissue reactions, consider prompt referral to their orthopaedic surgeon for review.
- The clinical association between MoM prostheses, Co/Cr ions in the blood and heart failure has not been established.
If you are treating a patient with another type of hip implant including another type of MoM implant, reassure the patient that the study found that - other than ASR XL- there was no evidence of a difference in rate of being hospitalised for heart failure between other MoM compared to MoP prostheses.
Patients with MoM implants should be followed up in accordance with the TGA's previous recommendations.
Patients with implants other than MoM should be followed up in accordance with standard clinical practice.
Information for orthopaedic surgeons
If you have implanted an ASR XL device, it is recommended that you consider advising patients of this issue during their next follow up visit.
To maximise the likelihood of early detection of adverse local tissue reactions, the recommendation for patients with large-head MoM implants (which includes ASR XL) is follow up on at least annual basis for the life of the implant. You may wish to consider measuring Co/Cr levels in the blood and imaging including MARS MRI.
Consideration should be given to referral of patients to their GP and/or cardiologist who may in turn wish to monitor those patients for early signs of heart failure, particularly if there are additional risk factors.
Such monitoring may include clinical examination and appropriate imaging such as echocardiogram.
If you have further questions or concerns regarding this issue, contact Johnson & Johnson Medical on 02 9815 4000 or 1800 252 194.
Background
As reported on the TGA website on 16 May 2011 the ASR total hip replacement system has been the subject of extensive international investigations.
This culminated in withdrawal of the implant from the Australian market in December 2009, and the worldwide recall of the implant in August 2010.
In 2016 the ASR implant has the highest revision rate of any hip implant used in Australia,[1] and the majority of the revisions are due to degradation of the tissue surrounding the implant caused by Co/Cr ions and debris generated by corrosion and wear of this MoM implant. It is well established that the Co/Cr ions and metallic debris can lead to adverse local tissue reactions, which are serious adverse outcomes in their own right.
The TGA has published information aimed at consumers and health practitioners about the ongoing care and follow-up of people with MoM implants including ASR, so that adverse local tissue reactions can be detected as early as possible.
There has also been a concern that the Co/Cr ions can enter the blood stream and that this may lead to adverse health effects due to systemic toxicity. A great deal of research has been devoted to determine if a link exists between MoM hip implants - particularly ASR - and systemic toxicity. The TGA has been monitoring international developments in this area. Although there have been some publications, the majority of these have been case studies involving single individuals and describe adverse health outcomes that have also been seen in people who have not had a MoM implant. Data from larger groups of patients are needed to confirm the possible connection between the adverse health effects reported in the case studies and having a MoM implant.
Details and findings of the Australian study
The study[2] was a retrospective cohort study conducted using the data in the Australian Government Department of Veterans' Affairs health claims database on patients who received conventional total hip arthroplasties (THA) between 1 January 2004 and 31 December 2012 as treatment for osteoarthritis.
A total of 4019 people were included in this study; 3.0% received an ASR XL prosthesis, 8.8% an alternative MoM prostheses and 88.2% received a MoP prosthesis. Analyses were performed for females and males separately. Men with ASR XL prostheses had a higher incidence of hospitalisation for heart failure after their hip replacement procedure compared with men who had MoP prostheses (15.9% compared to 7.6% respectively). The rate of being hospitalised for heart failure in men was more than three times higher for ASR XL compared to MoP prostheses (hazard ratio = 3.21; lower 95% confidence limit = 1.59; upper 95% confidence limit = 6.47). There was no evidence of a difference in rate of being hospitalised for heart failure between the other MoM prostheses compared with MoP prostheses in men (other large head (>32mm) MoM, 8.1% and small head (≤32mm) MoM, 5.3%). No difference in heart failure hospitalisation rates was found for women between the prostheses types. There was no significant difference in adjusted mortality rates by patients with different bearing surfaces in either men or women.
The authors conclude that: 'An association between ASR XL and hospitalisation for heart failure was found in men. While causality between ASR XL and heart failure could not be established in this study, it highlights an urgent need for further studies to investigate the possibility of systemic effects associated with MoM THA.'
Limitations of the study
The analysis conducted by the group has been thorough, but there are limitations that arise from the underlying features of the data available which may limit its use across all populations of MoM hip replacement patients:
- There were only 121ASR XL recipients (63 men, 58 women) in the cohort. This is considered a small number for a clinical study looking for differences in outcomes that have a low occurrence such as this; nonetheless the differences in the rate of hospitalisation for heart failure between the men with an ASR XL implant and the men with MoP implants was statistically significant.
- The median age in the study's cohort (82 years; range 38-99 years) was higher than that of all ASR XL and other MoM implant recipients in the general Australian population (median=67 years; range 13-103 years).[3] It is not clear what implications this difference may have for younger patients.
- The data did not include records of Co/Cr metal ion levels in the blood as this data was not available. This study found an association between having an ASR XL implant and a higher incidence of hospitalisation due to heart failure, but high Co/Cr levels from MoM hip prostheses leading to heart failure is speculation that still needs to be tested.
It is important to note that the study did not report a statistically significant difference in the rate of death between the ASR XL and the MoP group. Also there is currently no evidence to show that women with ASR XL implants are at a higher risk of hospitalisation due to heart failure.
Another important finding is that the risk of hospitalisation due to heart failure among people who had MoM hip implants other than ASR XL was not found to be significantly higher than that of those who had MoP implants.
References
- Information from the Australian Orthopaedics Association National Joint Replacement Registry (AOANJRR)
- Marianne H Gillam, Nicole L Pratt, Maria C S Inacio, Elizabeth E Roughead, Sepehr Shakib, Stephen J Nicholls & Stephen E Graves (2016): Heart failure after conventional metal-on-metal hip replacements, Acta Orthopaedica, DOI: 10.1080/17453674.2016.1246276
- Supplementary Report, 2015: 'Metal on Metal Bearing Surface Total Conventional Hip Arthroplasty' - (available from https://aoanjrr.dmac.adelaide.edu.au - this information is updated in October each year)
Reporting problems
Consumers and health professionals are encouraged to report problems with medical devices. Your report will contribute to the TGA's monitoring of these products. For more information see the TGA Incident Reporting and Investigation Scheme (IRIS).
The TGA cannot give advice about an individual's medical condition. You are strongly encouraged to talk with a health professional if you are concerned about a possible adverse event associated with a medical device.

